CHE 1302 Lecture Notes - Lecture 7: Stoichiometry, Hydrofluoric Acid, Rice Chart
Document Summary
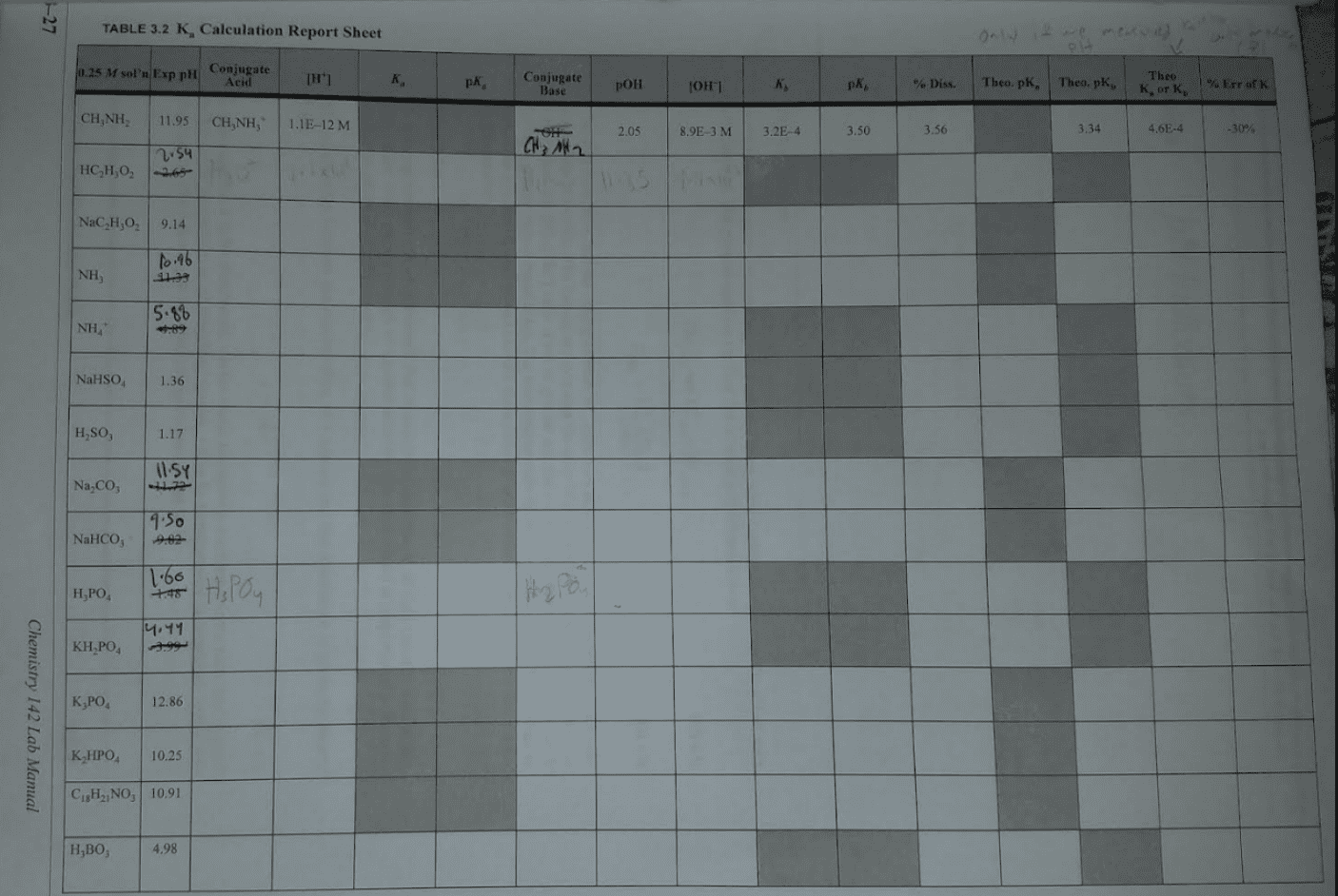
First we need a balanced equation and the expression for kb conjugate base conjugate acid base acid. Remember that h2o does not get a column in the table because it doesn"t appear in the expression for kb. We"ll take the initial [ch3nh2] to be 2. 40 m (this is sometimes called the nominal concentration ), and then we allow the base to ionize. Hence, the concentration of the molecular base will decrease, while concentration of its conjugate acid will increase, and concentration of oh will also increase. Now substitute equilibrium concentrations into the kb expression . We want to know ph. x = [oh ] = 0. 032 m poh = log(0. 032) = 1. 49 ph = 14. 00 poh = 12. 51. A similar procedure is used when determining the ph of a solution of a weak acid, given concentration and ka: What is the ph of a 1. 40 m solution of the weak acid hf? (ka = 3. 5 10 4)