CHEM 1A Lecture Notes - Lecture 8: Kinetic Theory Of Gases, Ideal Gas, Ambient Pressure
27 views5 pages
Document Summary
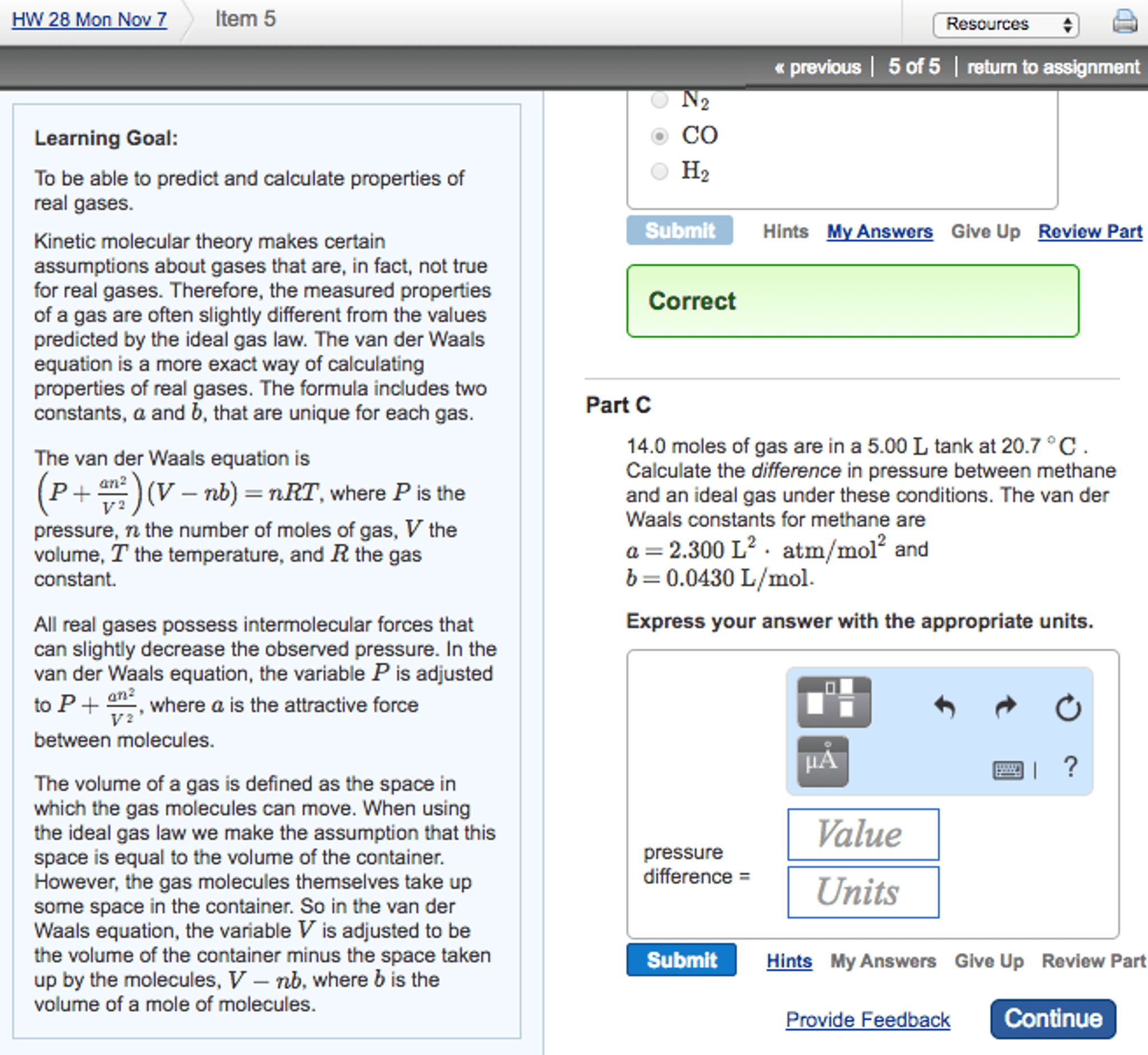
Gases, whether trapped in various containers or in the open atmosphere, are literally all around us. We know this when we measure pressure of a gas in a container the force exerted by the gas on the walls of the container per unit surface area. Macroscopic properties such as temperature and pressure all derive from the random motion of gas molecules as described by the kinetic molecular theory of gases. The most remarkable observation is that to a first approximation, all gases, regardless of their identity, behave the same way in response to changes in volume, pressure, temperature, and the number of gas molecules. Explain the properties of gases in terms of the random motion of molecules. Relate volume, temperature, and pressure of a gas sample in both rigid and flexible containers. Explain why the pressure, temperature, and volume of an ideal gas do not depend on the identity of the gas.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232