CHEM 1A Lecture Notes - Lecture 24: Make It Or Break It, Bond-Dissociation Energy, Activation Energy
Document Summary
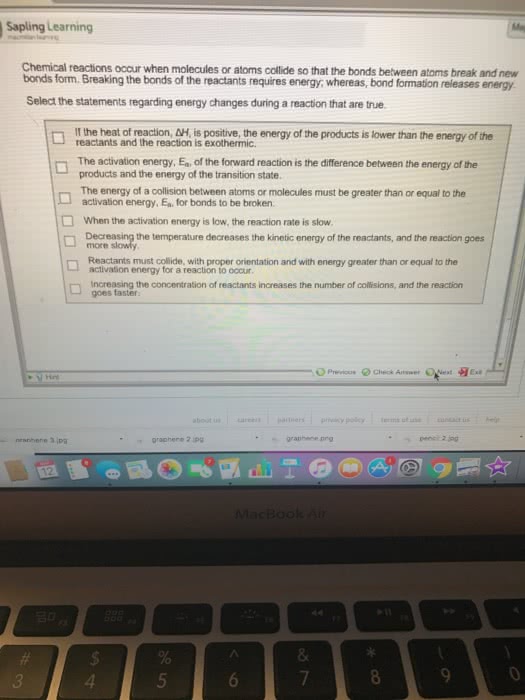
Methane, ch4, reacts with oxygen, o2, to form carbon dioxide, co2, and water, h2o. The co2 and h2o products have a high kineic energy, and are thus at a high temperature. The increase in kineic energy is due to a decrease in potenial energy. If more energy is released in forming bonds in the products than is required to break bonds in the reactants, then the reacion is exothermic. Average bond enthalpies can be used to predict the heat of reacions. It is convenient because then we can easily tell when potenial energy is negaive because of atomic interacions: why does the reacion of 2h + 2o . The reacion releases energy because the energy of forming the h-h and o=o bonds is negaive, meaning that energy is released in the form of energy. Explain this in terms of the balance between potenial and kineic energies of the reacing molecules. 2h2o, a large amount of energy is realized.