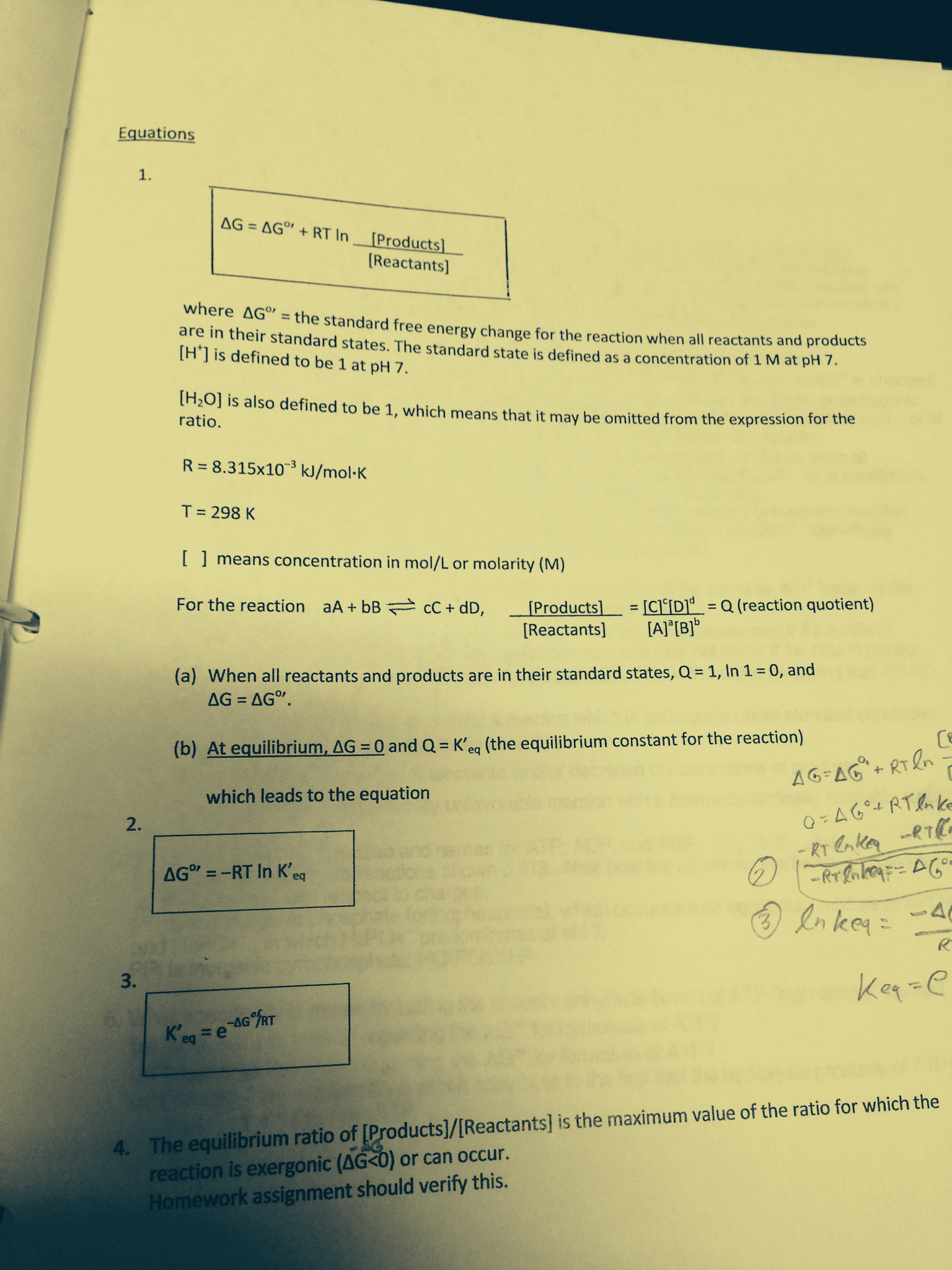CHEM 1A Lecture Notes - Lecture 28: Vapor Pressure, Reaction Quotient, Equilibrium Constant
71 views5 pages
Document Summary
The magnitude of negaive g expresses the driving force of a reacion to proceed towards products. As the reacion progresses, g becomes less and less negaive and reaches zero when the reacion reaches the state of equilibrium. If the selected iniial condiions yield g > 0, everything works the same except the reacion proceeds in the reverse direcion from products towards reactants unil it reaches the state of equilibrium. In all these cases, g can be related to the reacion quoient, q: Since at equilibrium g = 0 and q = k, it is easy to show that. G = --rt ln k. note that g and k are directly related to each other. Reacions with a negaive value for g have large values of k; those with posiive values of g have values for k smaller than 1. The general relaionship between delta g values of reacion and the k constant is that.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232