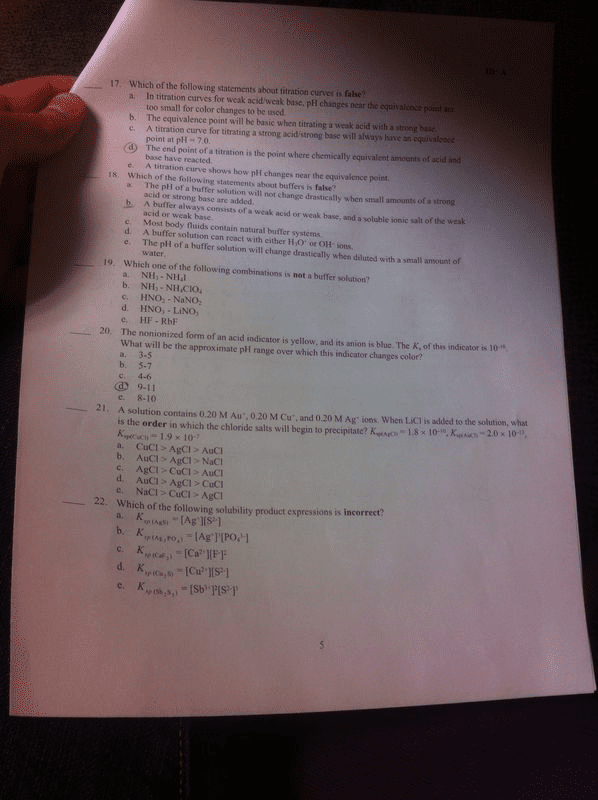CAS CH 102 Lecture Notes - Lecture 26: Conjugate Acid, Electrochemistry, Acid Rain
Document Summary
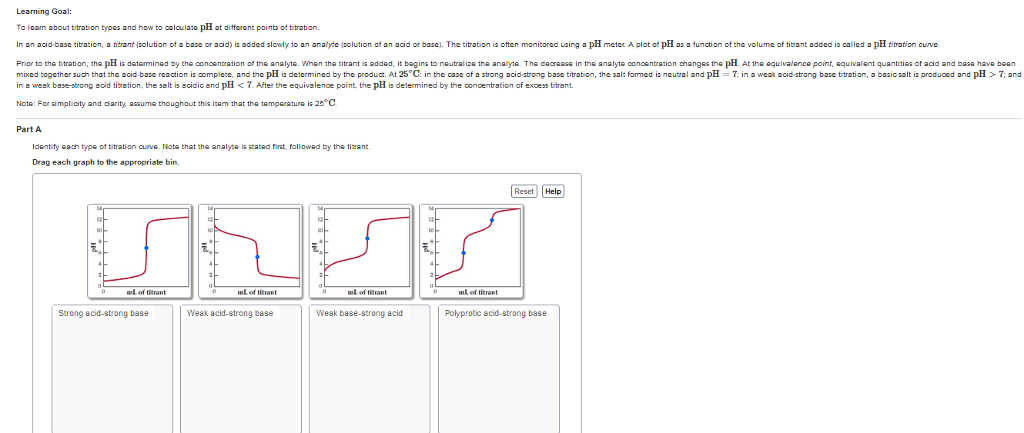
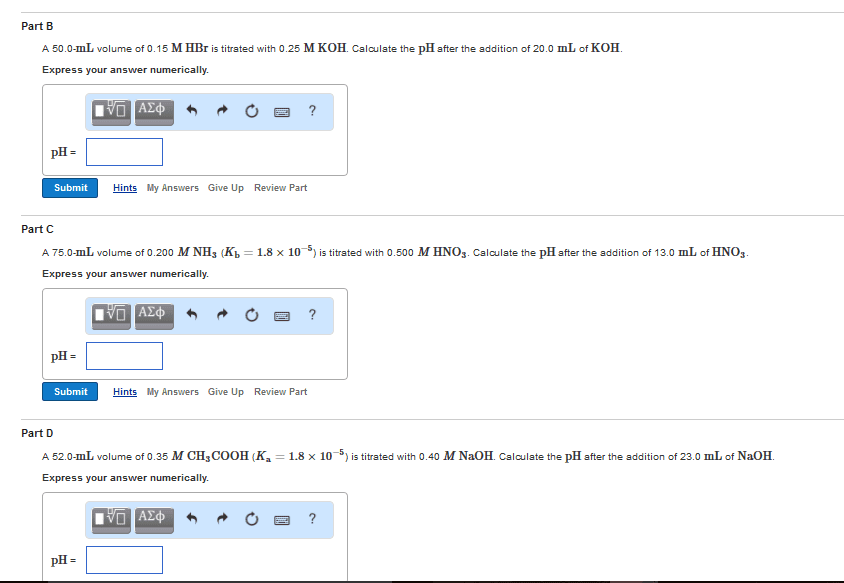
Conjugate acid base pairs: to identify cabp, the difference between the pairs is one h+ ion, presence or absence of one proton (h+, pairs. The pairs can predict the outcome of a reaction. Ph calculations: ph = -log[h+] ph = -log[concentration of h+ ions] Titration: use of volume measurement to determine concentrations of unknown solutions: equivalence point: concentration of h+ = oh- Solution is neutral: end point: when color change occurs. Does not necessarily mean solution is neutral. Buffer solutions: equilibrium systems that resist changes and maintain constant ph when acids or bases are added to it. Salts: ionic compound containing positive and negative ions: salt preparation methods. Correct acids and bases form that salt and water. Reaction of metallic oxide with nonmetallic oxide. Amphoteric substance: can act as both an acid or a base because it is neutral; can both accept or donate protons.