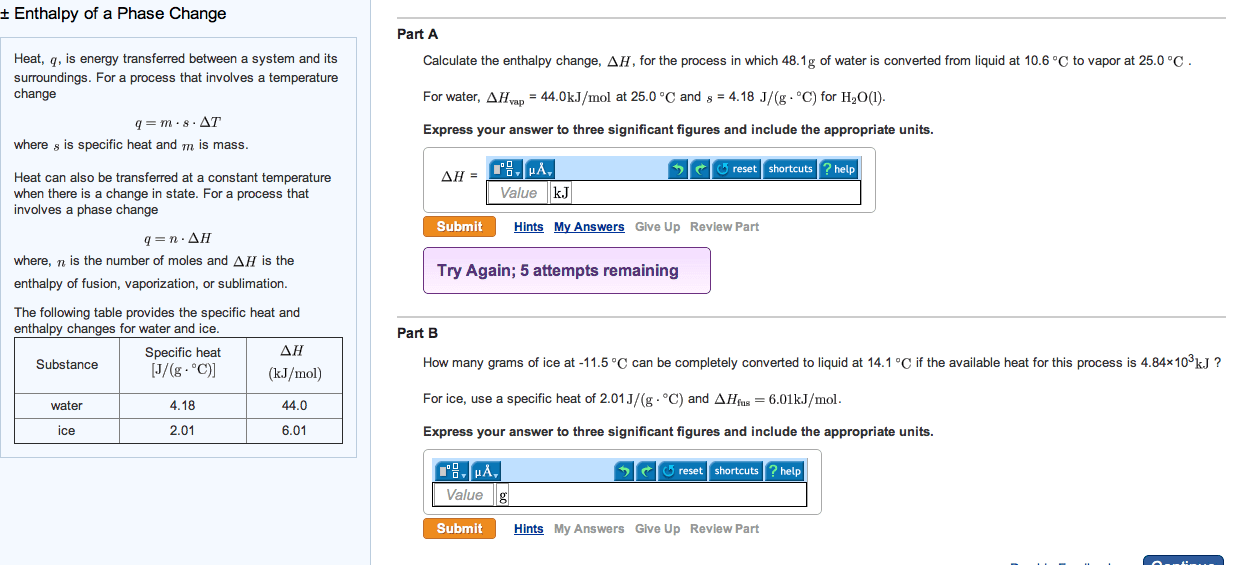
Enthalpy of a Phase Change Heat, q, is energy transferred between a system and its surroundings. For a process that involves a temperature change q = m middot 8 middot Delta T where s is specific heat and m is mass. Heat can also be transferred at a constant temperature when there is a change in state. For a process that involves a phase change q = n middot Delta H where, n is the number of moles and Delta H is the enthalpy of fusion, vaporization, or sublimation. The following table provides the specific heat and enthalpy changes for water and ice. Calculate the enthalpy change, Delta H, for the process in which 48.1g of water is converted from liquid at 10.6 degree C to vapor at 25.0 degree C For water, Delta Hvap = 44.0kJ/mol at 25.0 degree C and s = 4.18 J/(g middot degree C) for H2O(1). Express your answer to three significant figures and include the appropriate units. How many grams of ice at -11.5 degree C can be completely converted to liquid at 14.1 degree C if the available heat for this process is 4.84 Times 103kJ ? For ice, use a specific heat of 2.01 J/(g middot degree C) and Delta Hfus = 6.01kJ/mol. Express your answer to three significant figures and include the appropriate units.



