CAS CH 101 Lecture Notes - Lecture 6: Radiant Energy, Uncertainty Principle, Spectroscopy
Document Summary
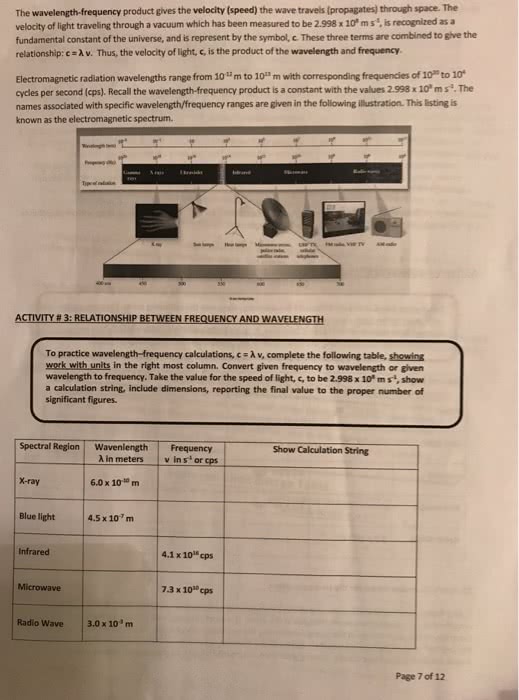
Radiant energy that exhibits wavelength-like behavior and travels through space at the speed of light in a vacuum. Wavelength: distance between two peaks in a wave. Frequency: number of waves per second that pass a given point in space. !e = change in energy, in j h = planck"s constant, 6. 626 ! E = energy m = mass c = speed of light. The laser light of a cd player has a wavelength of 7. 80 x. Continuous spectrum: contains all the wavelengths of light. Line spectrum: contains only some of the wavelengths of light. proved that only certain energies are allowed for the electron in the hydrogen atom. Spectroscopy is the study of electromagnetic radiation emitted or absorbed by a given chemical species. If light is shown through a solution of unknown concentration, the amount of absorbance can be used to calculate the concentration of that solution. A = absorbance a = molar absorptivity (l/mol cm)