I did the calculation part of it ( bottom right heading calculation ) but I was told they are wrong. Can someone please help me with the calculation paper of this report? The result of the reaction is given.
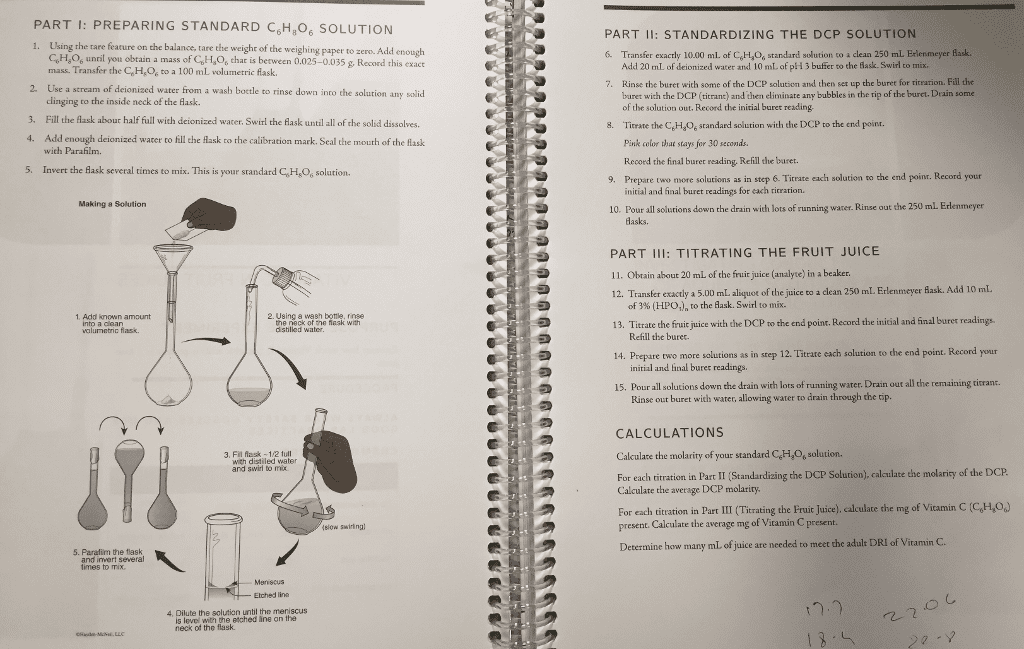

PART PREPARING STANDARD CSH o SOLUTION g the rare feature on the balance, tare the weighr of the weighing paper to zero. Add enough mass of CoHsoo chal between 0.025-0.035 g Record this exact mass. Transfer the CeH.Coe to a 100 mL volumetric flask. 2, Use a stream of deionized water from a wash boetle to rinse clinging to the inside neck of the Aask. 3. Fill the flask abour half full with de ized water, swirl the flask until all of the solid dissolves. 4. Add enough deionized water to till the flask to rhe calibration mark. Seal the mouth of the flask with Parafilm. 5. Invert the flask several times to mix. This is your standard CsHsoo solution. Making a Solution 2. Using a wal bottle, rinse Add known a the neck or the Mask with into a clea led 3, Fil flask with distilled water and swirl to mix. dow swirlingl the flask and invert several imes to mix, 4, the solution until the men scus is level with etched ine neck of the flask. PART STANDARDIZING THE DCP SOLUTION Transfer exactly 10.00 ml. of C6Haoo standard solution to a clean 250 mL Erlenmeye zed water and 10 mL of pH 3 buffer to the Bask-swirl Add 20 mL of dei Fill the se tbe buret with some of the DCP salution and n set up buret with the DCP (titrant) and then eliminate any bubbles in the rip of the buret.Drain some of the solution out.Record the initial buret reading, 8. Titrate the CEHaoestandard solution with the DCP to the end point. Pink color that stays for 30 seconds Record the final buret reading, Refill the buret. solutions as in step 6. Titrate each solution to the end poinr. Record your 9. Prepare two more nitial and final buret readings for each ritration. 10. Pour all solutions down the drain with lots of running water. Rinse out the 250 ml Erlenmeyer TITRATING THE FRUIT JUICE PART 11. obtain about 20 mL of the frui juice analyte) in a beaker 12. Transfer exactly a 5.00 mLaliquot of the juice to a clean 250 mm. Erlenmeyer Aask. Add 10 mL of 3% (HPO o the Mask. Swirl to mi 13. Titrate the fruit juice with the DCP to the end point. Record the initial and final buret readings. Refill the buret. 14. prepare two more solutions as in step 12. Tittate each so ion to the end point. Record your initial and hnal buret reading ing water. Drain out all the remaining tirrant. utions down the drain with 15, Pour all through the tip Rinse out buret with water, allowing water to d CALCULATIONS Calculate the molarity of your standard CeHao6 solution. of the DCP lculate the molarity Standardizing the DCP Solution ach titration in Part Calculate the average DCP molarity. For each titration in Part III (Titrating the Fruit Juice), calculate the mg of vitamin C (CoHeo6 present. Calculate the average mg of Vitamin C present. Determine how many mLof uice are needed to meet the adult DRI of Vitamin C





