CHEM 102 Lecture Notes - Lecture 5: Triethylamine, Reagent, Mesylate
27 views2 pages
Document Summary
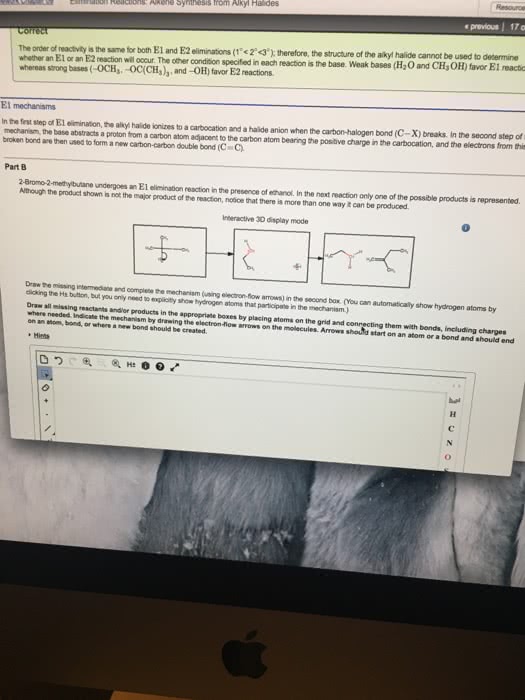
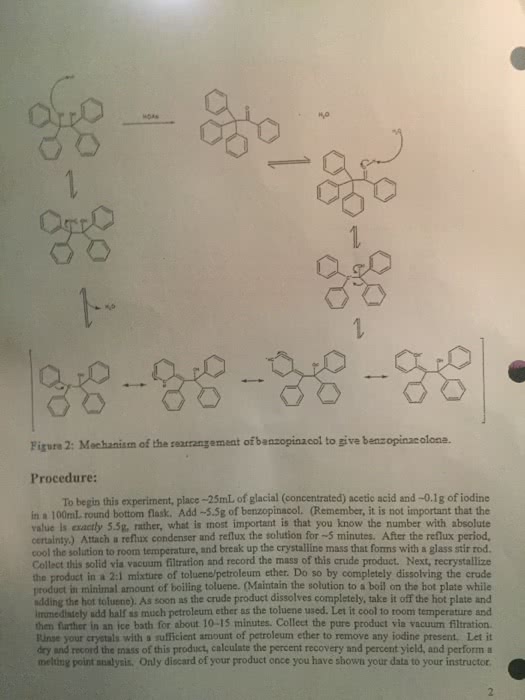
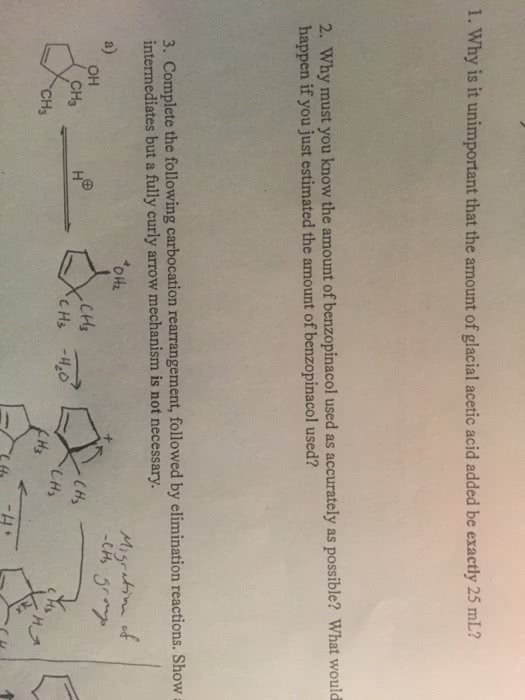
We haven"t yet been very adventurous with our choice of leaving groups for eliminations: all you have seen so far are e2 from alkyl halides and e1 from protonated alcohols. This is deliberate: the vast majority of the two classes of eliminations use one of these two types of starting materials. Since the leaving group is involved in the rate-determining step of both e1 and e2, in general, any good leaving group will lead to a fast elimination. You may, for example, see amines acting as leaving groups in eliminations of quaternary ammonium salts. In the second, a stabilized tertiary cation could be formed (so either e1 or e2 might occur), but no strong base is present, so the mechanism must be e1. You have just seen that hydroxyl groups can be turned into good leaving groups in acid, but this is only useful for substrates that can react by e1 elimination.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232