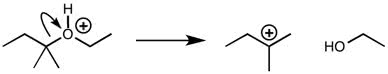
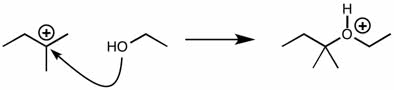
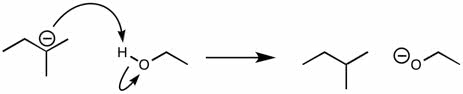
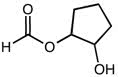
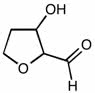
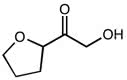
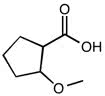
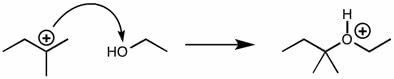CHEM 111 Lecture Notes - Lecture 19: Paramagnetism, Linear Combination Of Atomic Orbitals, Dihydrogen Cation
Document Summary
Diatomic molecular orbital theory mixing two atomic s orbitals, you get one bonding orbital and one antibonding orbital. Note that bonding orbitals can have nodes (usually at the nuclei) In-phase = orbitals overlap, create a longer, stretched version of the two orbitals together. Out-of-phase = one orbital is shaded out, nodal plane. 2 p orbitals out-of-phase = pi* s & p orbitals in-phase = sigma sp s & p orbitals out-of-phase = sigma sp * limit to electrons in orbitals - 2. always 2, spin-paired if there are two. Homo of one atom moves into the lumo of another to make an orbital. Bo = bond order (# of bonding - # of antibonding)/2. 2 electrons in h2, h2 = -1 electron (1 - 0(no antibonding))/2. Mixing 5 atomic orbitals makes the same number of molecular orbitals. 2 bonding and 2 antibonding this makes sense - helium doesn"t bond with anything. Overlap is most important for orbitals with similar energies.