CHEM 127 Lecture 4: Quantum Mechanics Lecture
636 views4 pages
Document Summary
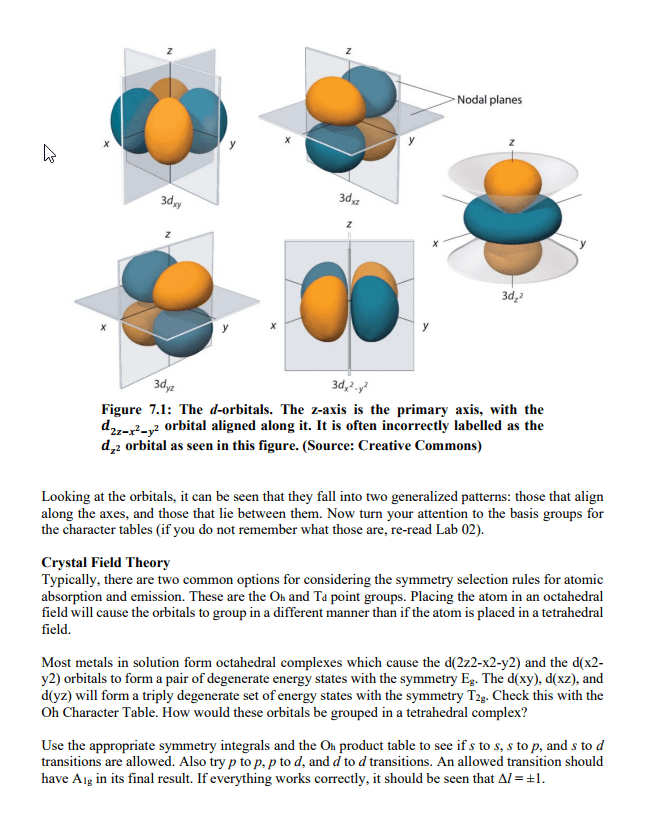
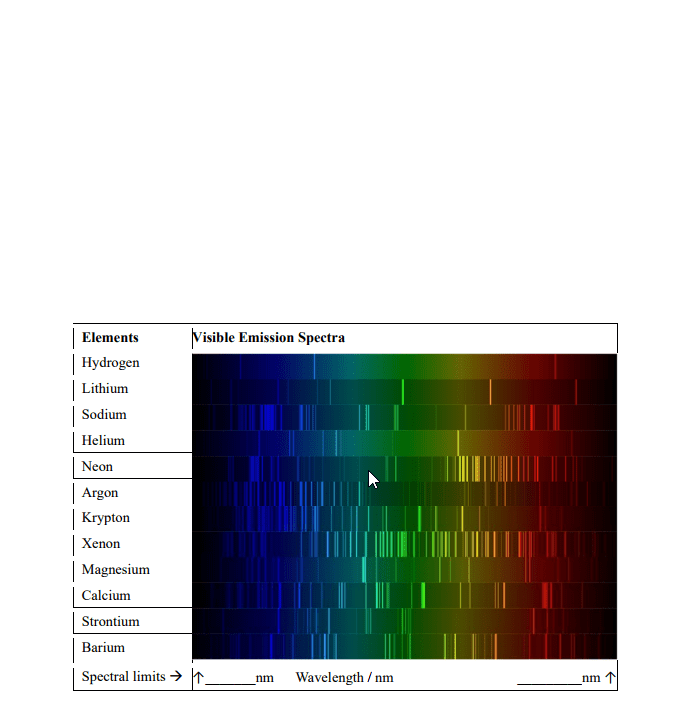
The energy difference between two energy levels in a sodium atom is 3. 38 x 10^-19, so a photon with this energy is emitted when an electron moves from the higher to the lower energy level. Calculate the wavelength of light corresponding to this electron transition. Explain why every atom has a unique emission spectrum. It is because every atom has different sizes of energy levels that the electrons would have to jump. The bohr model only works if you have one electron. Only was capable of describing an atom with one electron. Allows for the description of atoms with more than one electron. Waves in water show interference patterns as the waves interact with each other. Similar things happen with light when it is shined through closely spaced slits. Thought that since light can behave like a particle, can light also behave like a wave since some particles have wave like attributes.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Calculus
4 Edition,
Rogawski
ISBN: 9781319050733
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Single Variable Calculus: Early Transcendentals
4th Edition, 2018
Stewart
ISBN: 9781337687805
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
CALCULUS:EARLY TRANSCENDENTALS
4 Edition,
Rogawski
ISBN: 9781319050740
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232