Classify each statement about effective nuclear charge, Zeff, as true or false.
In a Be atom, a 1s electron has a greater Zeff than a 2s electron.
Across a period, as Zeff increases, atomic size decreases.
Electrons in a p orbital are more effective than those in other orbitals at shielding other electrons from the nuclear charge.
A 1s electron is a Be atom has a smaller Zeff than a 1s electron in a Li atom.
Effective nuclear charge decreases from left to right across a period on the periodic table.
Effective nuclear charge is dependent on the number of electrons present in an atom.
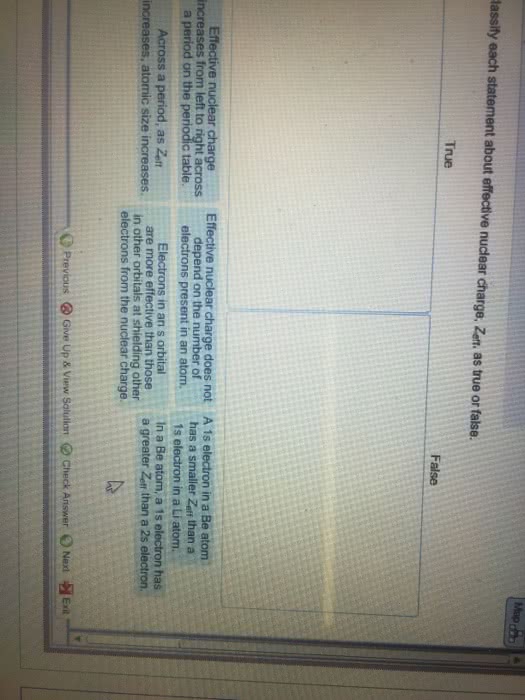
Classify each statement about effective nuclear charge, Z_eff, as true or false. In a Be atom, a 1s electron has a greater Z_eff than a 2s electron. Electrons in a p orbital are more effective than those in other orbitals at shielding other electrons from the nuclear charge. Effective nuclear charge decreases from left to right across a period on the periodic table. Across a period, as Z_eff increases, atomic size decreases. A 1s electron In a Be atom has a smaller Z_eff than a 1 s electron in a Li atom. Effective nuclear charge is dependent on the number of electrons present in an atom.