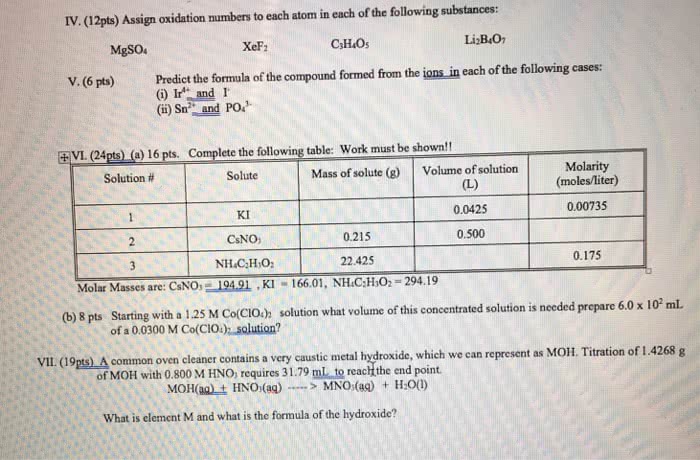
CHEM 090 Lecture Notes - Lecture 5: Lead, Sodium Hydroxide

Chemistry 90 Chapter 5 Lecture Notes
• Synthesis reactions: 2 or more compounds react to form one product
o Ex: 2H2(g) + O2(g) → 2H2O(g)
• Decomposition reactions: single reactant is converted to 2 or more simpler substances
o Ex: 2Ag2CO3 → 4Ag + 2CO2 + O2
• Combustion reactions: substance reacts w/ oxygen and proceeds w/ the evolution of
heat, producing carbon dioxide and water
o Ex: CxHyOz + O2 → CO2 + H2O
• Single replacement reactions: one element within a compound is replaced by another
element, usually in an aqueous solution
o The replaced atom is lower in activity series than the replacing atoms
o Ex: Zn(s) + H2SO4(aq) → H2(g) +ZnSO4(aq)
• Double replacement reactions: 2 compounds exchange partners with each other to form
2 new compounds
o Ex: Pb(NO3)2 + 2HCl → PbCl2 +2HNO3
• Solubility Rules:
o Most nitrate salts are soluble
o Most salts of Na, K, and NH4 are soluble
o Most chloride salts are soluble, except AgCl, PbCl2, Hg2, Cl2
o Most sulfate salts are soluble, except BaSO4, PbSO4, CaSO4
o Most hydroxide compounds are insoluble, except NaOH, KOH, Ba(OH)2, Ca(OH)2
o Most sulfide, carbonate and phosphate salts are insoluble
find more resources at oneclass.com
find more resources at oneclass.com