CHM-1045 Lecture Notes - Lecture 10: Mean Free Path, Kinetic Theory Of Gases, Exothermic Reaction
Document Summary
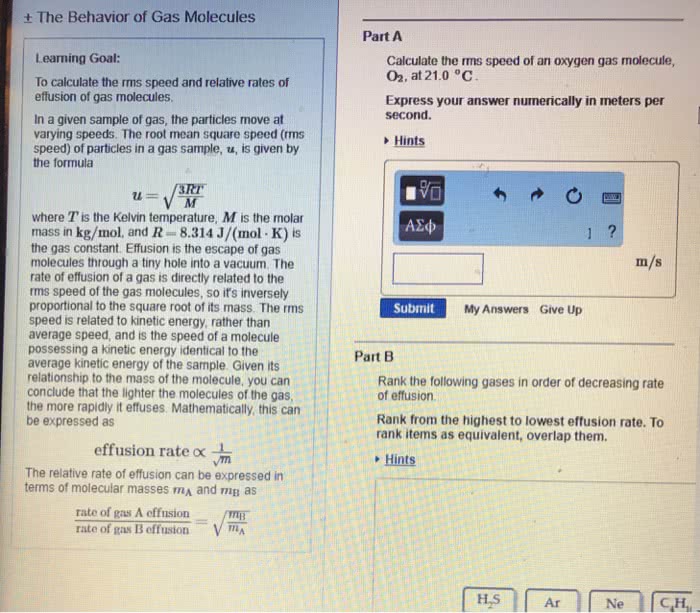
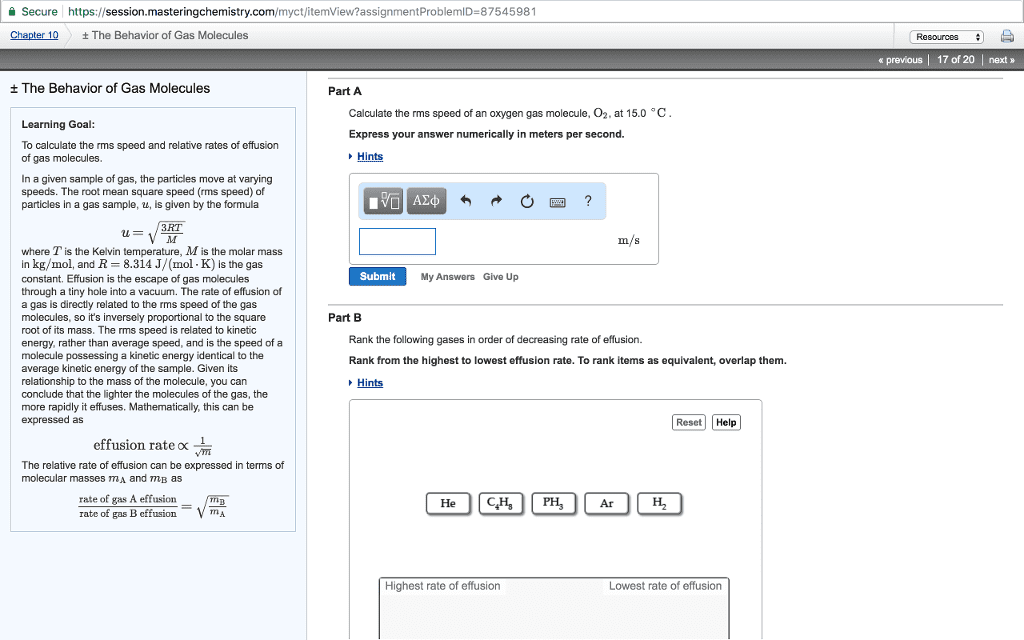
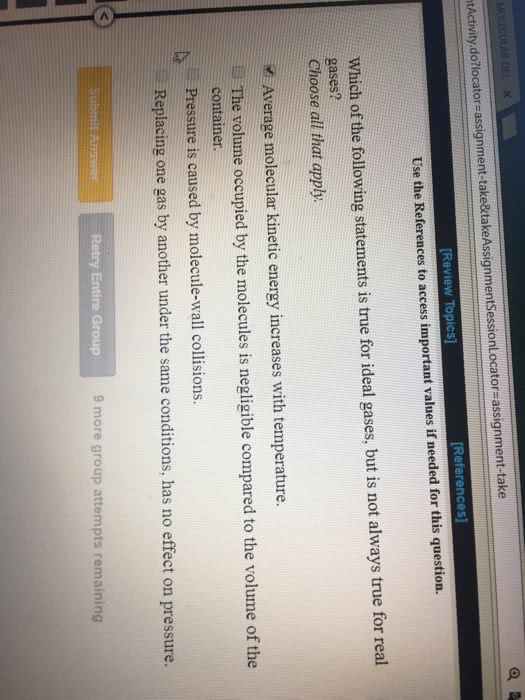
The kinetic molecular theory: gases molecule travel in continuous motion. Gas are elastic: the average kinetic energy of a gas is proportional to its kelvin temperature. Diffusion: when atom move from high concentration to low concentration. Effusion: is when molecule escape through a hole. Rate of effusion increase with smaller mass, and high temperature. Mean free path: average distance a molecule travel between collision. The mean path increases with decreasing pressure. Rate of effusion: (cid:2869) where m is the molar mass of the gas. Graham"s law of effusion: (cid:2869) (cid:2870)= (cid:2870) (cid:2869) using this equation when using two gas. Capacity to do work and supply heat. Kinetic energy: the energy an object process while it is in motion. Potential energy: energy an object process while it is in position. Law of energy: energy can change form, but never created nor destroyed. Thermal energy: energy of molecule in random motion. Exothermic reaction: is when the system release heat.