CHEM-002 Lecture Notes - Lecture 21: Cella, Oxidation State, Electrochemistry
43 views2 pages
25 Mar 2017
School
Department
Course
Professor
Document Summary
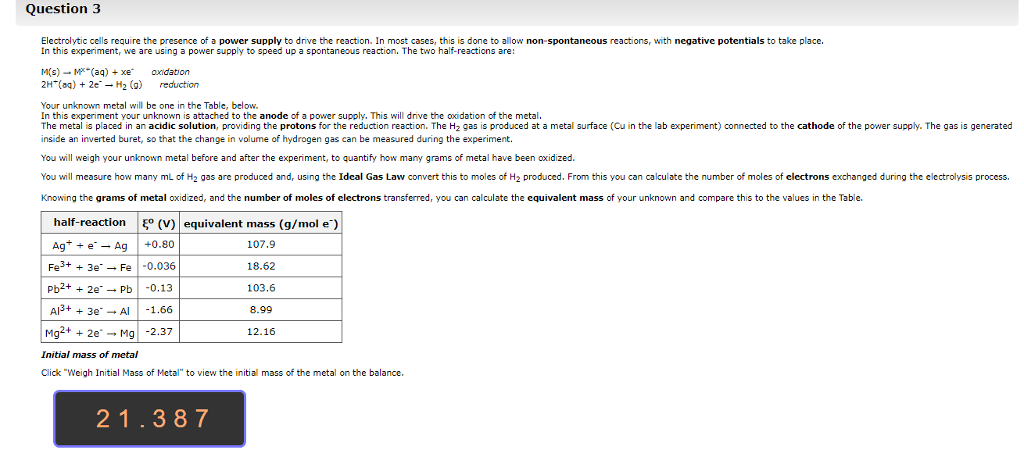
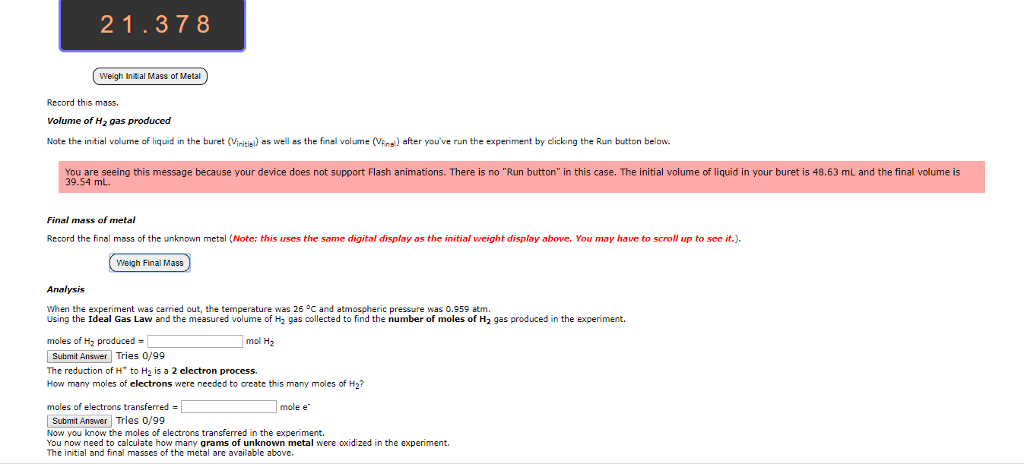
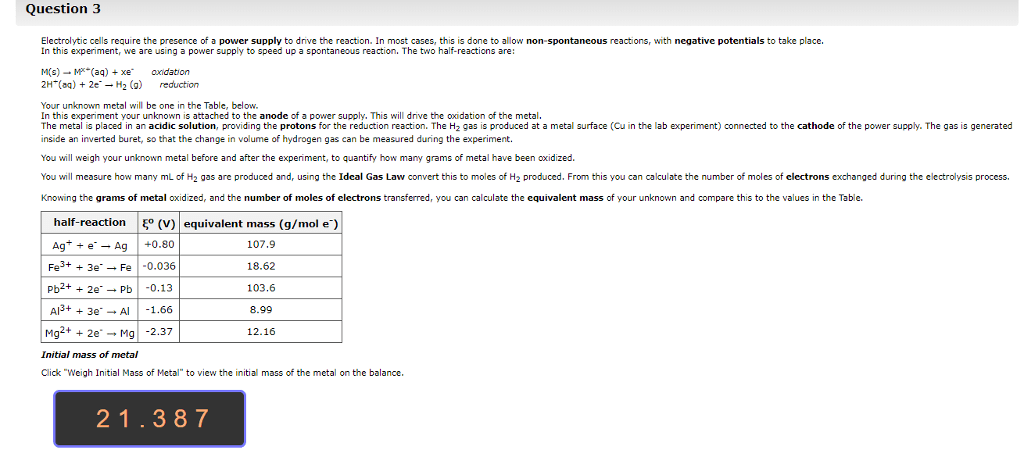
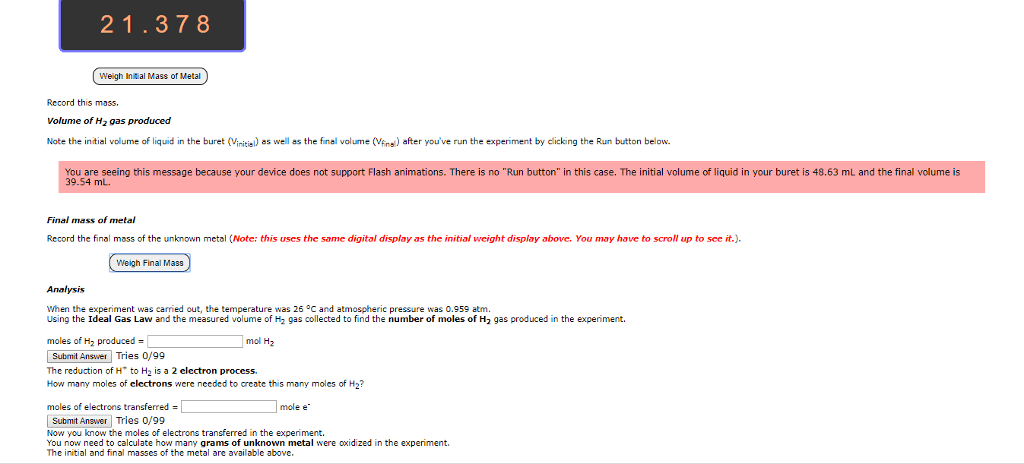
Cells often need to perform reactions that are not spontaneous for example, tying amino acids together to make proteins. Sometimes a cell will combine a non-spontaneous reaction with a spontaneous reaction. The most common spontaneous reaction is that atp splits and this provides the energy for the non- spontaneous reaction so that can actually happen. When there is oxidation there must be reduction because electrons have to go somewhere. To understand electrochemistry, we need to be able to keep track of electrons, which we do by assigning oxidation numbers. The higher the oxidation number, the fewer electrons the atom has. There are a bunch of rules that control this, and you have to prioritize them to figure out what to do. Sum of the oxidation numbers in a neutral molecule must be equal to zero. Flouride has an oxidation number of -1. Alkali atoms get an oxidation number of -1.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232