Chemistry CHEM S-20ab Lecture Notes - Lecture 13: Lindlar Catalyst, Alkene, Alkyne

Chemistry E-2a: Lecture 9 December 11, 2008
1
Test Yourself Now!
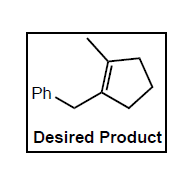
Provide a complete synthesis of the desired product from the indicated starting material. You may use
any organic or inorganic reagents in your synthesis.
OH
HO
OH
Chemistry S-20ab
Week 3
1


Chemistry E-2a: Lecture 10 December 18, 2008
2
Nomenclature of Alkynes
• Draw skeletal structures for:
acetylene
dimethylacetylene
1-butyne
2-butyne
propargyl chloride
Reading: Section 14.1
Chemistry S-20ab
Week 3
17
Document Summary
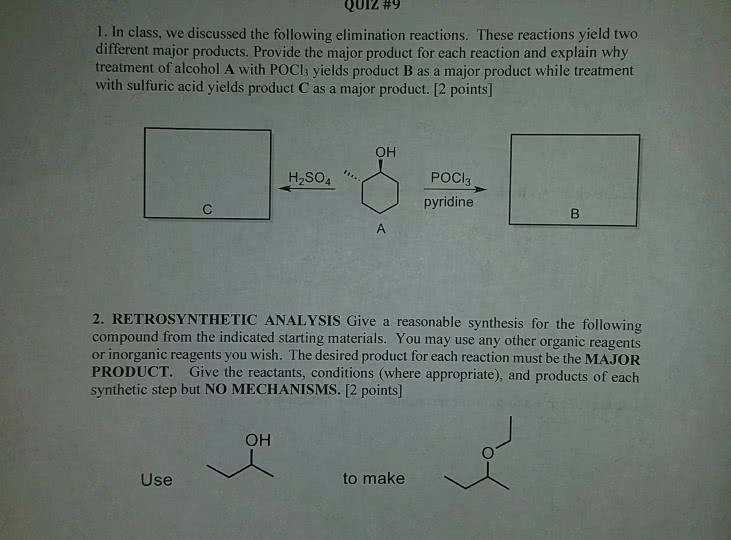
Provide a complete synthesis of the desired product from the indicated starting material. You may use any organic or inorganic reagents in your synthesis. Predict the product and draw a complete curved-arrow mechanism for the following reaction: Draw a complete curved-arrow mechanism for the following reaction: Predict the product and draw a complete curved-arrow mechanism for the following reaction. Be sure to show the stereochemistry of the product! The enol is not the final product of the reaction of an alkyne with water. The enol reacts rapidly with the aqueous acid to form a carbonyl compound: in this case, a ketone. Show the complete synthetic transformation from an alkyne to a ketone: The following reaction produces an unstable intermediate known as an enol (alkene + alcohol = enol). Draw a complete curved-arrow mechanism for this reaction up to the formation of the enol. You should also explain why the alkene geometry is trans (that is, you should explain the stereochemistry!)