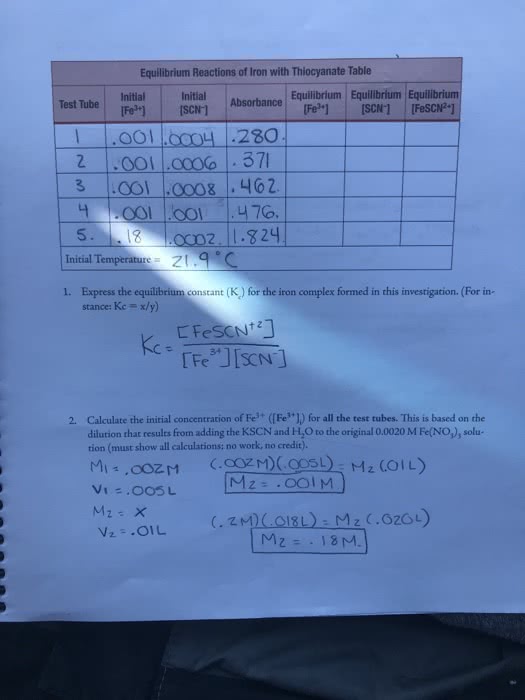
CHEM-C 243 Lecture Notes - Lecture 13: Molar Concentration, Stoichiometry, Silver Chloride
Document Summary
Section 1- the equilibrium condition: chemical equilibrium- state where the concentrations of all reactants and products remain constant with time. Concentrations reach levels where rate of forward reaction equals rate of reverse reaction. Has nothing to do with amounts of each product and reactant. On molecular level, there is frantic activity (dynamic, not static situation) In most cases, products revert back to reactants (reverse reaction) and those will react again in ongoing process. In stoichiometry we talk about theoretical yields, and the many reasons actual yields may be lower. Make enough cookies to stock a shop. Do not have to replace the numbers of cookies right away to keep stock up, but the rate of cookies being produced must equal the rate at which they are being sold. Stock stays constant but does not have to be full (100% yield); reached equilibrium. 80%); making too many cookies can also disrupt: as a reaction progresses: