030.102 Lecture Notes - Lecture 3: Acid Dissociation Constant, Phenolphthalein, Titration
Document Summary
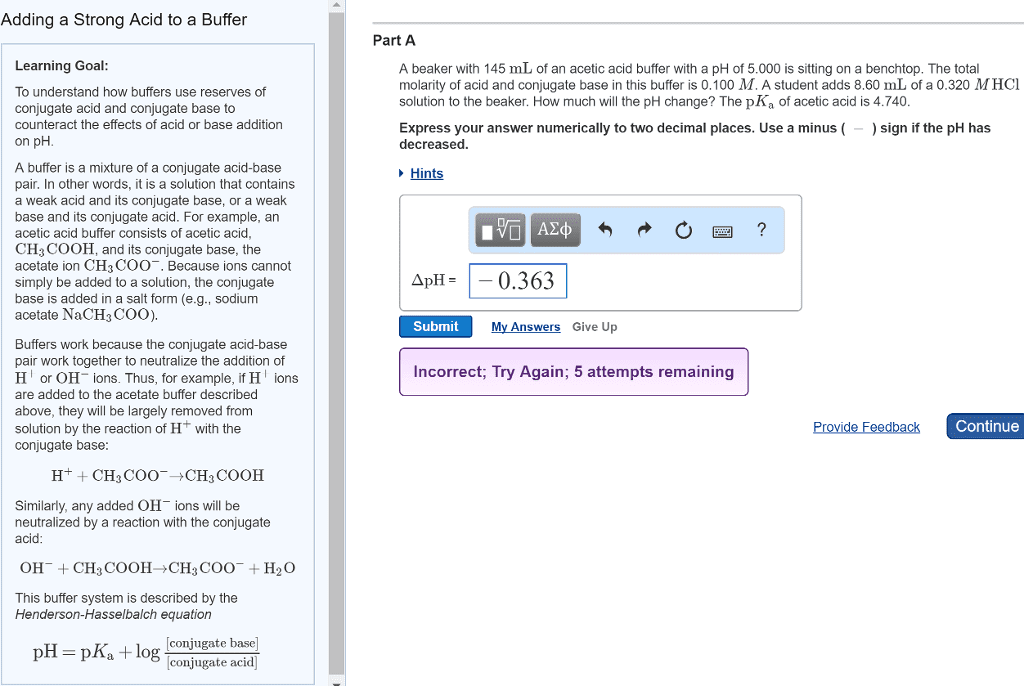
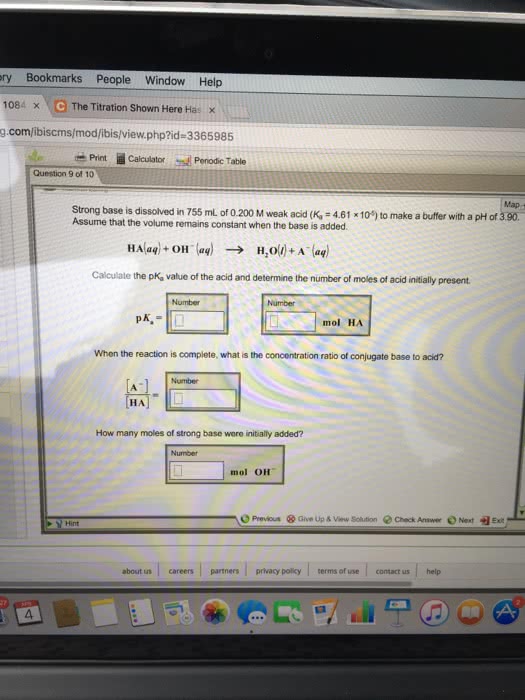
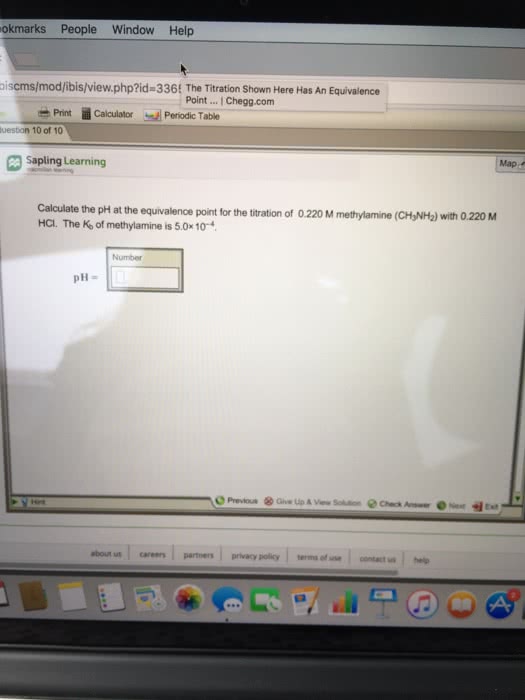
Henderson-hasselbalch equation o ph = pka log [acid]/[conj. Concentration of h3o+ depends on ratio of weak acid and conjugate base. Must have concentration of ha and a- to be nearly equal and large compared to any acid or base added to solution. Technique uses stoichiometry to determine concentration of unknown solution. Indicators are chemicals that are added to reaction to determine when it is complete. Reaction form is acid + base salt + water. Salt = cation from base + anion from acid. Example: h2so4 + ca(oh)2 caso4 + 2h2o. Procedure used to determine the concentration of an acid or base. Base solution is titrant in buret o o o. Color changes when there is no longer excess acid present, when base is added, it forms water, when the salt forms the color changes. Endpoint: just enough base has been added to neutralize all the acid.