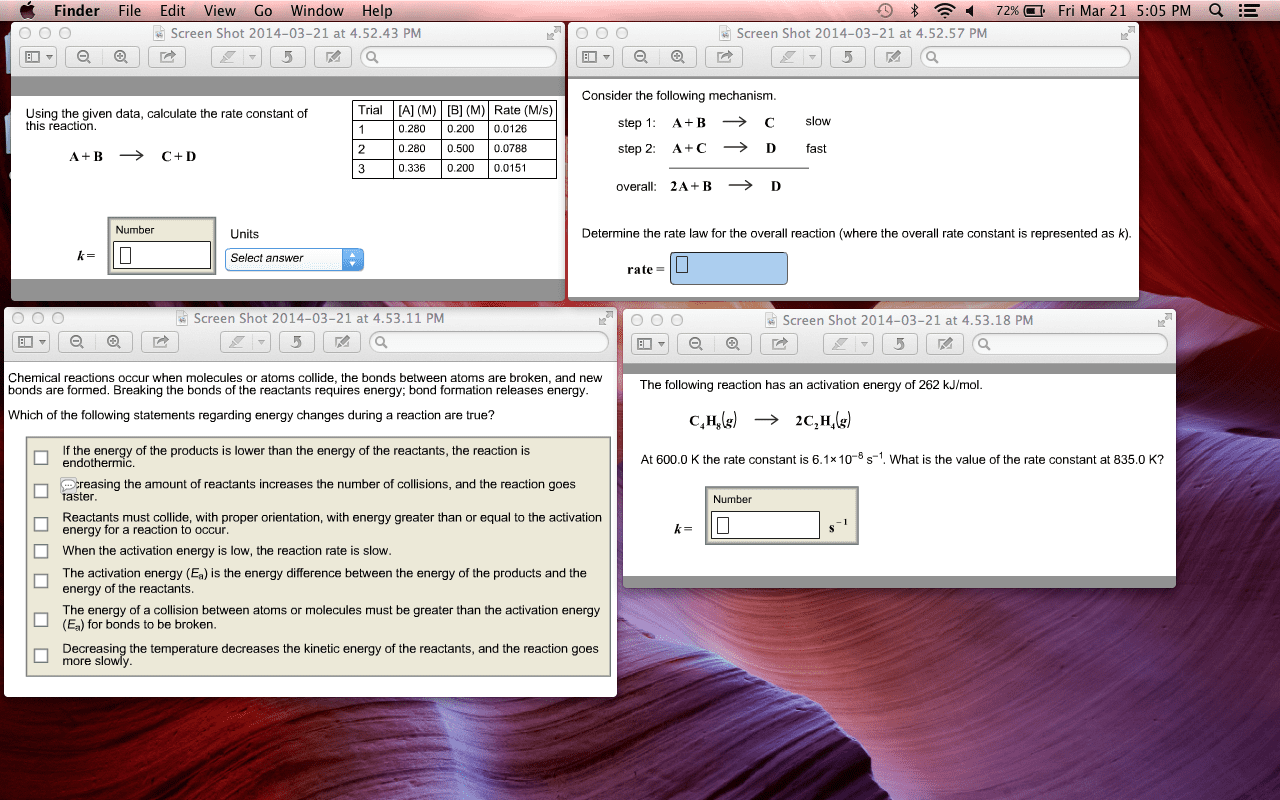
CHEM 1214 Lecture Notes - Lecture 1: Enthalpy, Collision Theory, Activated Complex
15 views2 pages
15 Sep 2016
School
Department
Course
Professor
Document Summary
Reaction rate: reaction rate: change in concentration over change in time, a b. Rate of disappearance of a: - [a]/ t: 2n2o5 4no2 + o2. (- [n2o5]/ t) = ( [no2]/ t) = 1/1( [o2]/ t: we cannot calculate a single number that expresses the rate for the entire reaction. Instead, we can calculate the average rate over a given interval of time (final-initial), or the instantaneous rate at a given instant in time (slope of tangent at specific time). Collision theory: reactions occur when molecules collide with sufficient kinetic energy to break chemical bonds. The reaction then proceeds and the activated complex splits into the products. Chemical kinetics: study of rate of change of concentrations of substances involved in chemical reactions. Reaction rate: how rapidly a reaction occurs; related to rates of change in concentration of reactants and products over time.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232