CHM 143 Lecture Notes - Lecture 17: Calorimeter, Calorimetry, Chemical Energy
Document Summary
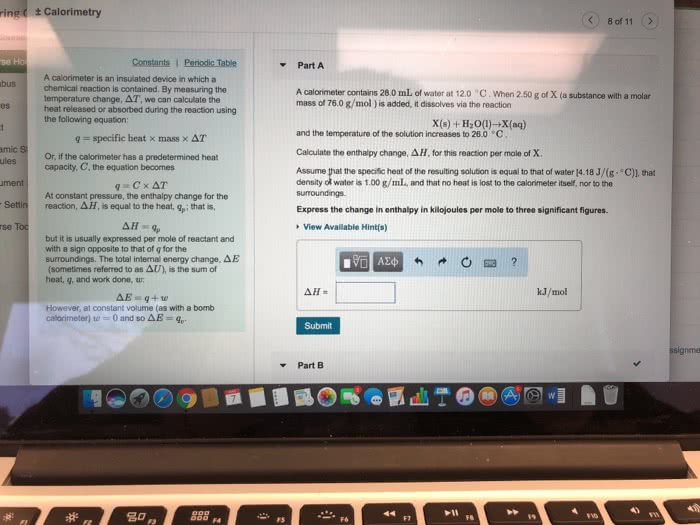
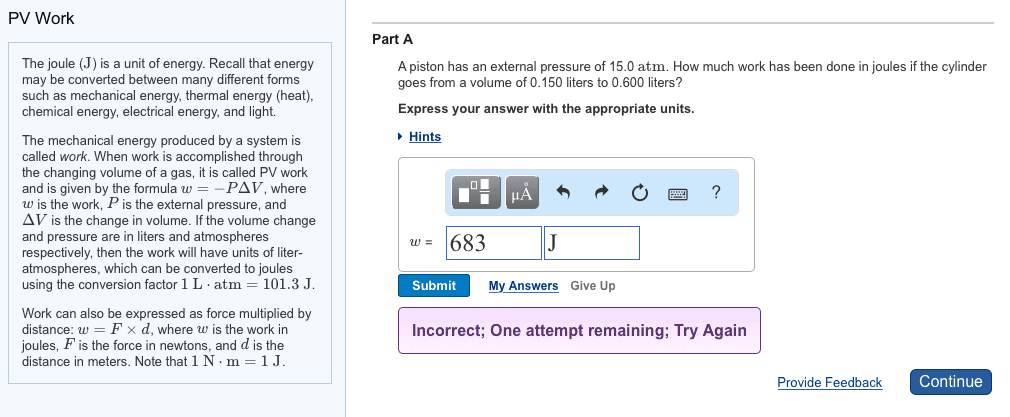
Atm * l w= -1. 02 atm(3. 8 l-2. 0l) w= -1. 84 atm x l x 101. 31j = -186 j. Constant volume calorimetry (bomb calorimetry) w= - p v. All energy that would be done as work (p v) gets converted to heat energy. No matter can enter or leave system. Highly insulated device so that no heat exchange to outside of calorimeter. Qsystem= - qsurrounding assume no heat lost to outside of calorimeter. Calorimeter has a heat capacity constant called calorimeter constant (ccal) units= cal/oc or j/oc. This constant is for the entire calorimeter therefore no mass or quantity. From before qcalorimeter= ccal x t (cid:932)(cid:934)(cid:931)(cid:931)(cid:928)(cid:934)(cid:927)(cid:917)i(cid:927)g(cid:932) (cid:930)= c x t (cid:930)= (cid:926) x cs x t. If qsystem is + then qsurroundings = - t= - If qsystem is - then qsurroundings = + t= + If qsurroundings = qcal then qreaction = -qcal qsystem = -qsurroundings qreaction.