METEO 300 Lecture Notes - Lecture 9: Specific Volume, National Institute Of Standards And Technology, Intensive And Extensive Properties
11 views2 pages
Document Summary
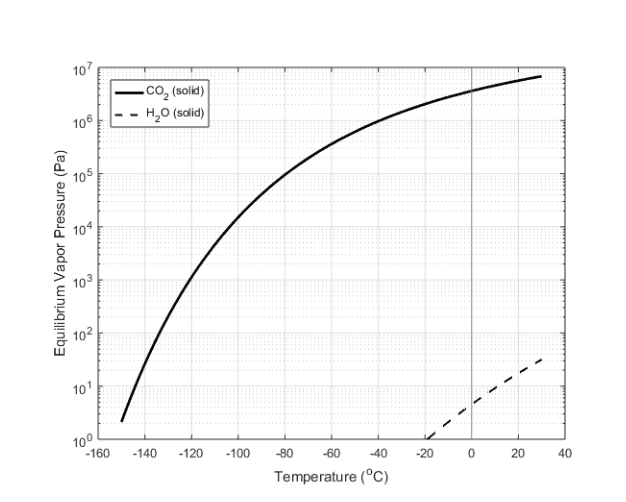
Me 300 - lecture 9 - properties of two-phase systems. Remember the state principle: for a single, compressible substance, the thermodynamic state is completely defined by specifying two independent intensive properties. You will be given two properties, and using a table, you will be able to determine the phase of the substance. After determining the phase, you will be able to find the unidentified property. How do you determine the phase of a non-ideal substance (water): you look at table d. 1 or d. 2 which gives you the values for the saturated temperatures. Using the nist webbook or appendix from the textbook, you should find that water is in the liquid phase in this example since the given temperature (360 k) is less than the saturation temperature (537. 09 k). Then, you will go to table d. 4 at a pressure of 5 mpa and find the specific volume corresponding to a temperature of 360 k.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers