CHM 11500 Lecture Notes - Lecture 8: Photon, Rydberg Constant, Rydberg Formula

27
CHM 11500 Full Course Notes
Verified Note
27 documents
Document Summary
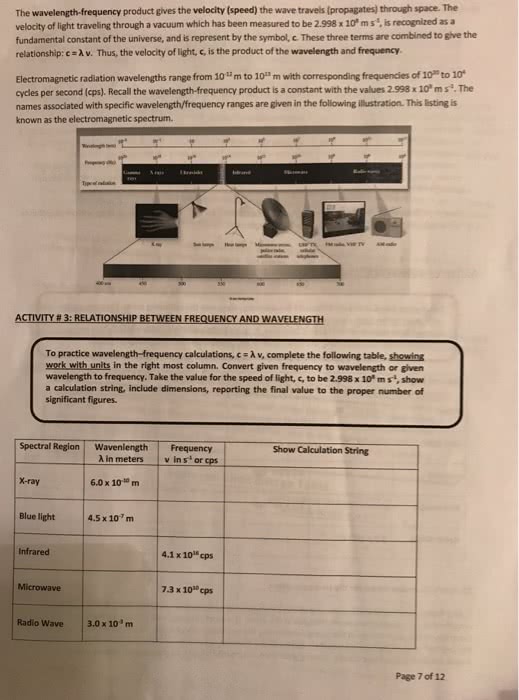
Chm 115 - lecture 8 - quantum theory. Wavelength ( ) - the distance a wave travels in one cycle. Frequency ( ) - the number of cycles a wave completes in one second. A has the longest wavelength and the lowest frequency. C has the shortest wavelength and the highest frequency. Wavelength and frequency are inversely proportional to one another. Amplitude - the amount of change that occurs in a wave over a single cycle. Waves with the same wavelengths/frequencies but different amplitudes: A has a larger amplitude than b. C = (the wavelength times the frequency of electromagnetic radiation is equal to the speed of light) C is equal to the speed of light in a vacuum: 3 * 108 m/s. When atoms become excited, the electrons are excited to higher energy levels. When excited electrons decrease in energy levels, to lower levels or their ground state, photons are emitted.