01:160:162 Lecture Notes - Lecture 7: Reaction Rate Constant, Activation Energy, Propene
Document Summary
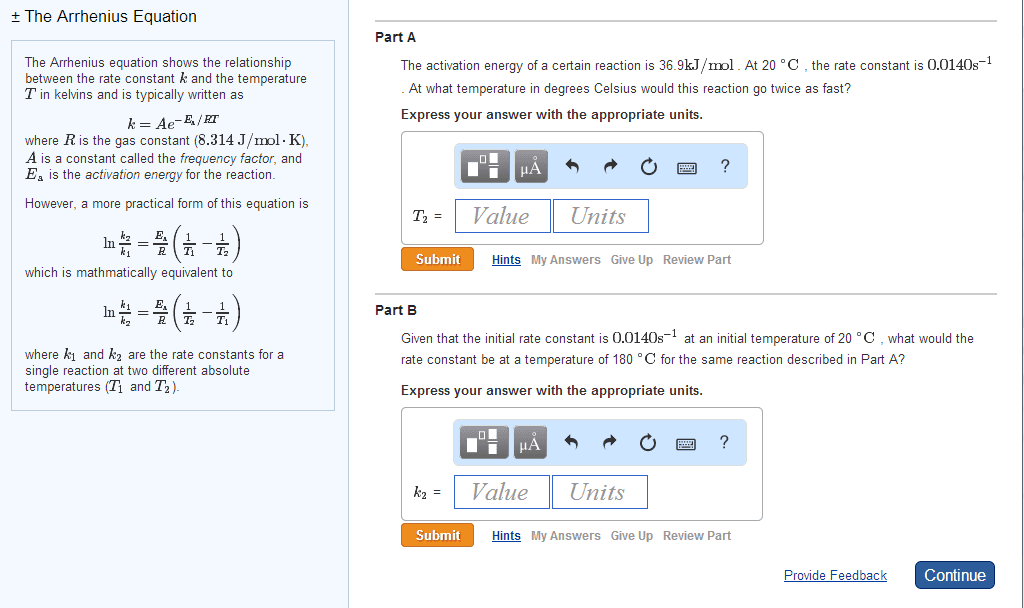
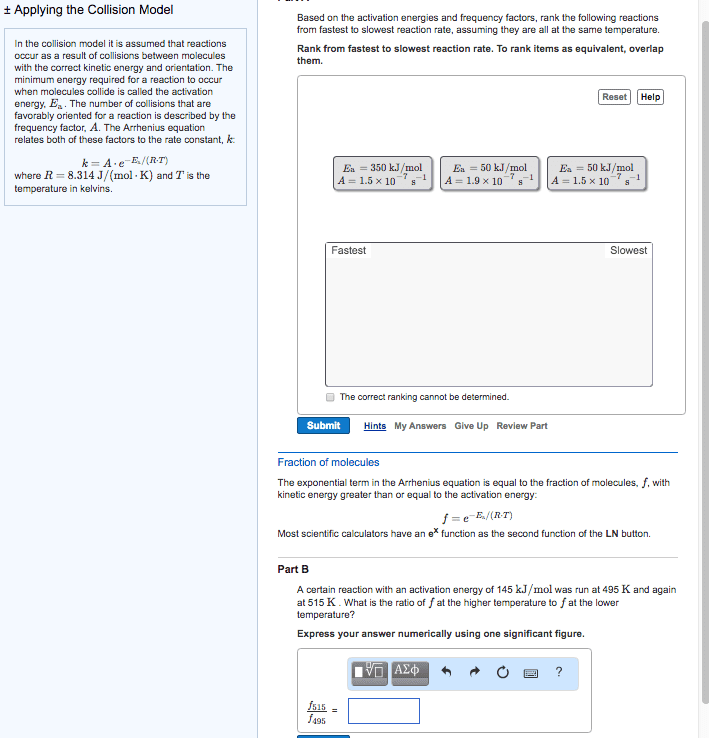
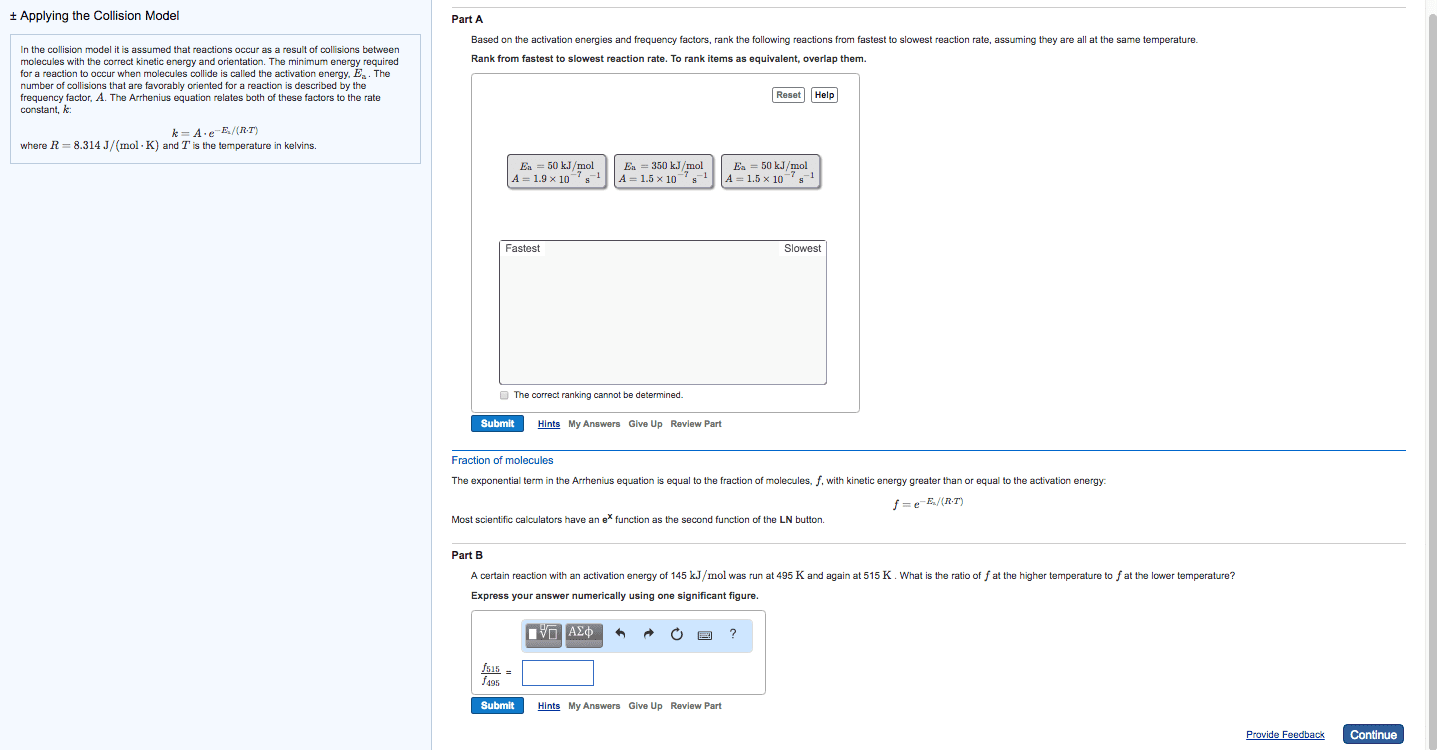
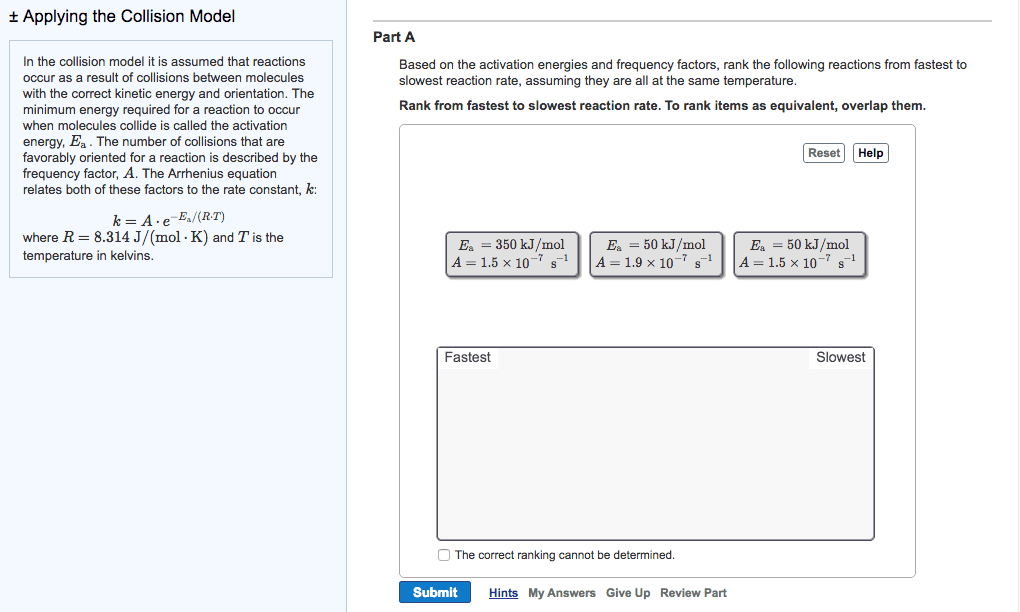
High rate constant = faster rate of reaction. Arrhenius equation mathematical relationships between magnitude of rate constant and other factors (ex. temperature, energy, etc. ) ** k = ae (-ea / rt) ** K = rate constant (units depend on reaction order) A = frequency factor (same units as k) Ea = activation energy (j / mol) Minimum amount of e needed for a reaction to take place. E needed to overcome activation barrier of a reaction. Ex. fuse on a stick of dynamite. Ea is always positive since e must be supplied / absorbed for reaction to proceed. Numerically large ea leads to a slow reaction. Numerically small ea leads to a fast reaction. Plot of relative change in e as reaction proceeds from reactants to products. Ea and overall reaction enthalpies can be determined. Frequency factor number of times a reactant approaches an activation barrier per unit time. Approaching an activation barrier is not the same as overcoming it.