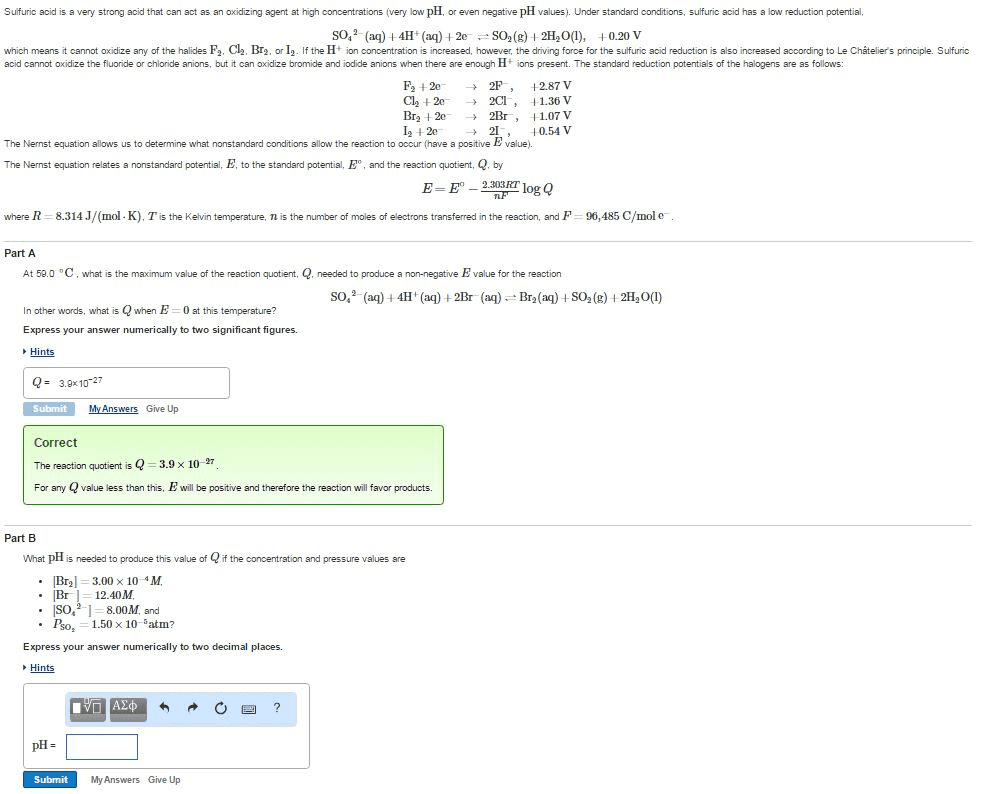
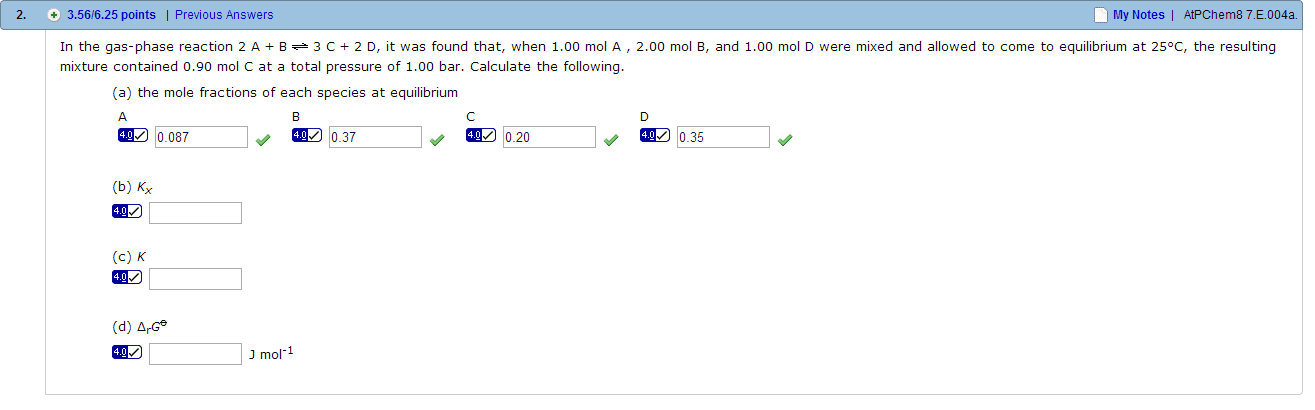
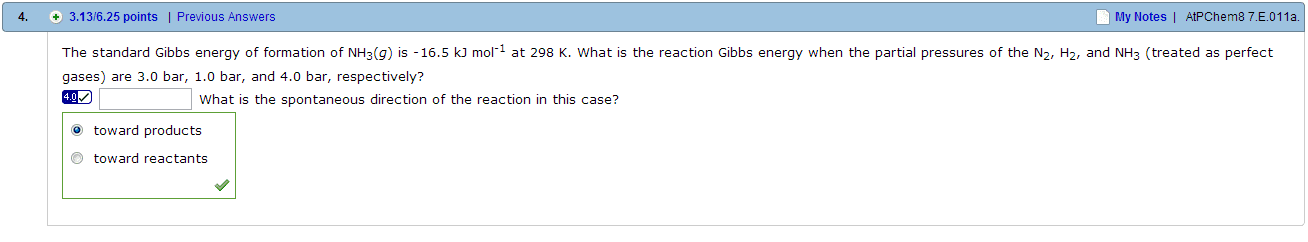
01:160:162 Lecture Notes - Lecture 29: Reaction Quotient, Water Vapor, Partial Pressure
Document Summary
Free e changes (cid:900)(cid:933)(cid:914)(cid:927)(cid:917)(cid:914)(cid:931)(cid:917) (cid:857) (cid:888) (cid:858) (cid:935)(cid:932) n(cid:928)(cid:927)(cid:932)(cid:933)(cid:914)(cid:927)(cid:917)(cid:914)(cid:931)(cid:917) (cid:857) (cid:888)(cid:858) c(cid:928)(cid:927)(cid:917)(cid:922)(cid:933)(cid:922)(cid:928)(cid:927)(cid:932) (cid:888) when all reactants and products are in their standard states. Partial pressure of each gas is 1 atm. Concentration of each dissolved species is 1 m. Partial pressures of gases are other than 1 atm. Concentrations of dissolved species are other than 1 m. At a given t there is only one way for standard conditions to occur but an infinite number of ways for nonstandard conditions to occur. Above scenario is not under standard conditions. Water vapor would have to be present at a pp of 1 atm along with pure liquid water present at 1 atm of external pressure. Nonstandard grxn needs to be calculated from standard grxn (cid:899)(cid:918)(cid:925)(cid:914)(cid:933)(cid:922)(cid:928)(cid:927)(cid:932)(cid:921)(cid:922)(cid:929) (cid:915)(cid:918)(cid:933)(cid:936)(cid:918)(cid:918)(cid:927) (cid:888) rxn (cid:914)(cid:927)(cid:917) (cid:888)rxn. Grxn = nonstandard free energy change (kj/mol) G rxn = standard free energy change (kj/mol) For reactions involving gases: q = qp. For reactions involving dissolved species q = qc.