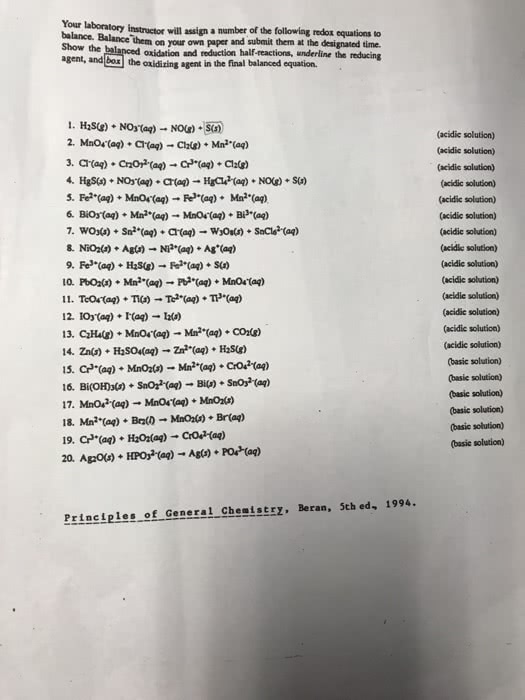
Your laboratory instructor will assign a number of the following redox equations to balance. Balance them on your own paper and submit them at the designated time. Show the balanced oxidation and reduction half-reactions, underline the reducing agent and box the oxidizing agent in the final balanced equation. DO 5-10
Your laboratory instructor will assign a number of the following redox equations to balance. Show the agent, and Balance 'them on your own paper and submit them at the designated time. oxidation and reduction half-reactions, underline the reducing the oxidizing agent in the final balanced equation. (acidic solution) (acidic solution) (acidic solution) (acidic solution) (acidic solution) (acidic solution) (acidic solution) (acidie solution) (acidie solution) (acidie solution) (acidie solution) (acidic solution) (acidic solution) (acidic solution) (basic solution) (basic solution) (basic solution) (basic solution) (basic solution) (basic solution) 10. Pots) * Mn2+(aq) â Pb-(aq) + MnO4-(ag) Prineiples of General Chemistry, Beran, 5th ed, 1994.