CHEM 1032 Lecture 16: 2/20/2017 Equilibrium; Mass Action Expressions; Equilibrium Constants
Document Summary
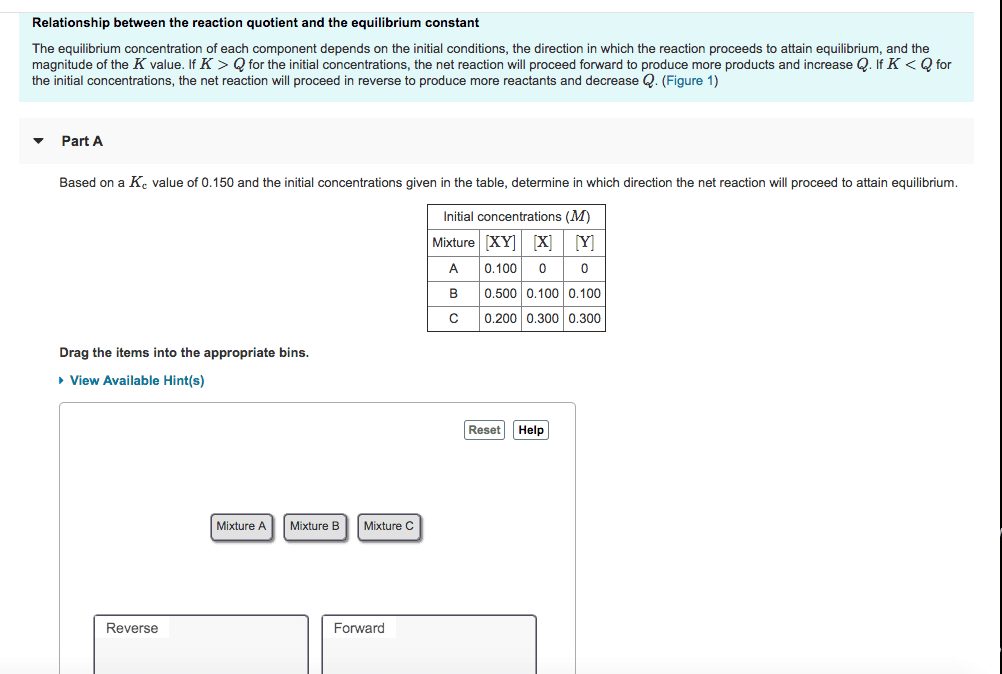
13. 1 chemical equilibria: equilibrium: forward and reverse reactions occur at equal rates, in a closed container, the products do not yield at 100, some reactants remain, at some point concentrations stop changing, reversible reaction (reverse arrows) All reactions can be reversible, but most, only under certain conditions: chemical equilibrium is a dynamic process, must differentiate between low reaction and actually equilibrium, rate of forward and backward reaction is the same. In dilute solutions, molarity and activity will be close to equal. Pure liquids and solids are not involved in equilibrium constant expressions. 1/k: kc and kp expressions, aqueous solutions, use kc in mol/liter, h2co3 (aq) > 2h+ (aq) + co3. 2-)/ (h2co3: gasses can use kc or kp, 2so. 3 (g) > 2so2 (g) + o2, kp = kc(rt)change(n, r= 0. 0206, change (n) is the difference between the moles of gas product and the. Do not include solids in the kc expression: [co2]/[co]2, hso4.