CH 101 Lecture Notes - Lecture 2: Conversion Of Units, Kinetic Energy, Exothermic Process
Document Summary
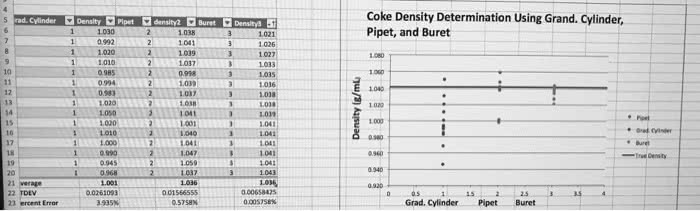
Section 2. 2: the reliability of a measurement depends on the instrument used to make the measurement, scientific measurements are reported so that every digit is certain except the last, which is estimated, accuracy. Refers to how close the measured value is to the actual value: precision. Refers to how close a series of measurements are to one another or how reproducible they are: random error. Error that has equal probability of being too high or too low: systematic error. Error that tends toward either being too high or too low. Ratio of mass (m) to volume (v) Independent of the amount of the substance (i. e. density: extensive property. Dependent on the amount of the substance (i. e. mass) Action of a force through a distance: kinetic energy (ke) Energy associated with motion: potential energy (pe) Energy associated with position: total energy = potential energy (pe) + kinetic energy (ke, thermal energy (te)