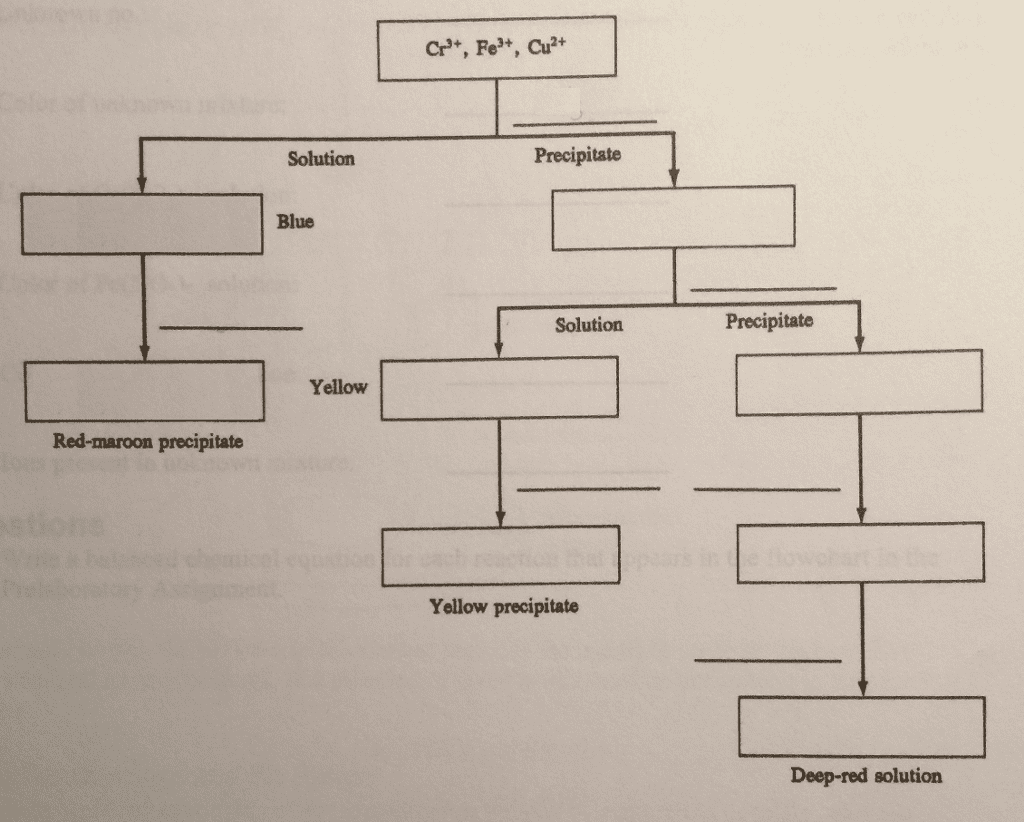
Complete the following flowchart by inserting the reagents and products from each reaction. Also show your work by showing the step-by-step reactions.

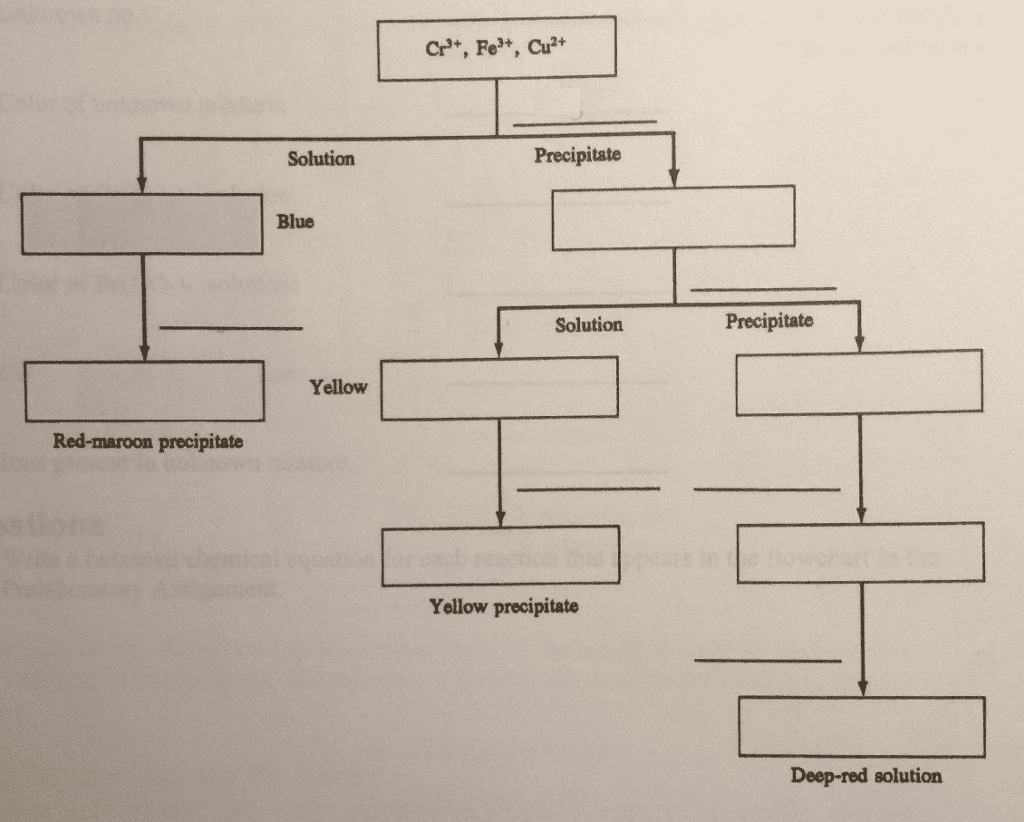
Purpose You will learn to separate mixtures of Cr3, Fe, and Cu2 and to identify each ion. Finally, you will be able to determine which of these ions are present in an unknown mixture. Concept of the Experiment Separation begins when aqueous ammonia is added to a mixture of the three ions. Because aqueous ammonia is a weak base, it is a source of ammonia as well as a source of hydroxide ions. As a consequence, this reagent causes the precipitation of the hydroxides of Cr3 and Fe . It also causes the formation of Cu(NH3), a deep blue complex ion, which remains in solution. Decanting (pouring off) the solution from the precipitate separates copper from chromium and iron. The presence of copper is confirmed when a red-maroon precipitate of Cu2Fe(CN)s forms after the addition of an acid (to neutralize NH3) and a solution of K4Fe(CN)6 To continue the separation, the hydroxides of Cr and Fe are treated with hydrogen peroxide in basic solution. Only Cr(OH) is oxidized. Yellow CrO (chromate ion) is formed in solution, whereas Fe(OH)s remains as a precipitate. When the solution is decanted from the precipitate, chromium and iron separate. The presence of chromium in the solution is confirmed when a yellow precipitate of PbCrOt forms after a solution of Pb(NO3)2 is added The precipitate of Fe(OH)s is then dissolved in an acid. The presence of iron is confirmed by the formation of a deep red color due to Fe(SCN), a complex ion, after the addition of a solution of KSCN In the Prelaboratory Assignment you will construct a flowchart for the entire separation scheme in order to become more familiar with it. You can use this scheme to separate and identify any combination of these metal ions in an unknown mixture. Also note that each of these metal ions has a characteristic color. As a result, you should use the color of your unknown mixture to support your analysis using the scheme.