CHE 2A Lecture Notes - Lecture 5: Isopropyl Alcohol, Chemical Formula, Molecular Mass
91 views5 pages

19
CHE 2A Full Course Notes
Verified Note
19 documents
Document Summary
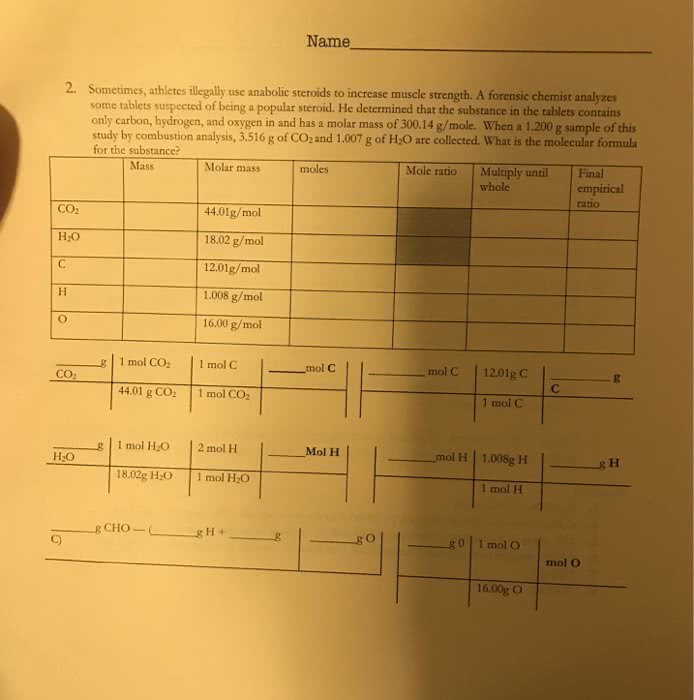
Chemistry 2a lecture 5 notes stoichiometry continued . Oct 12th, 2017, professor o. gulacar, uc davis: the empirical formula for adipic acid is c3h5o2. What is the molecular formula of adipic acid? (c: 12. 01g/mol; o: 16. 00g/mol; h. :1. 008g/mol : first find the empirical formula"s mass: c3h5o2. Next divide the sample"s mass by the empirical formula"s mass = 146/73. 07 = 1. 99 or 2. Therefore the actual formula has 2x the number of each type of atom in the empirical so. Process: find mass of empirical formula sample mass/empirical mass multiply that quotient with each atoms subscript in the empirical = compound"s actual formula. Combustion: usually involves hydrocarbon reactants and mainly produces co2 and h2o. To analyze it combustion reaction"s products are detected to determine various properties of the reaction: isopropyl alcohol, sold as rubbing alcohol, is composed of c, h, and o. 0. 255 g of isopropyl alcohol produces 0. 561g of co2 and 0. 306 g of h2o.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers