CHEM 001B Lecture Notes - Lecture 8: Kinetic Theory Of Gases, Torr, Partial Pressure
37 views3 pages
Document Summary
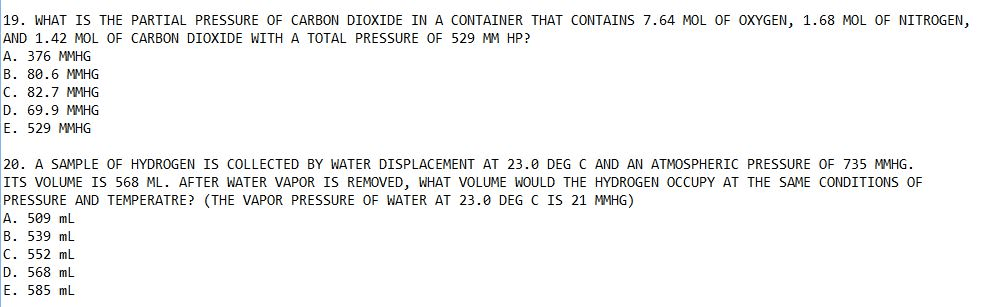
Chem 001b lecture 8: chapter 6. 8 - 6. 9. At 25 , a 1. 0 l flask contains 0. 030 mol of nitrogen, 150. 0 mg of oxygen, and 4 x 10 21 molecules of ammonia. Calculate the partial pressure and mole fraction of each gas. Nh 3 = (4. 21 x 10 21 molecules) ( N total = 0. 030 + 0. 00469 + 0. 00664 = 0. 0413 mol. 2 kclo 3 (s) 2 kcl (s) + 3 o 2 (g) When the water level inside the container matched the water level outside the container the pressure inside p total equals the atmospheric pressure p atm. A sample of kclo 3 is heated and decomposes to produce o 2 gas. The gas is collected by water displacement at 25 . The total volume of the collected gas is. 229 ml at a pressure of 754 torr. V = 229 ml = 0. 229 l. Correct for the partial pressure of water:
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232