CHM 2046C Lecture Notes - Lecture 9: Faraday Constant, Thermodynamics, Sodium Chloride
Document Summary
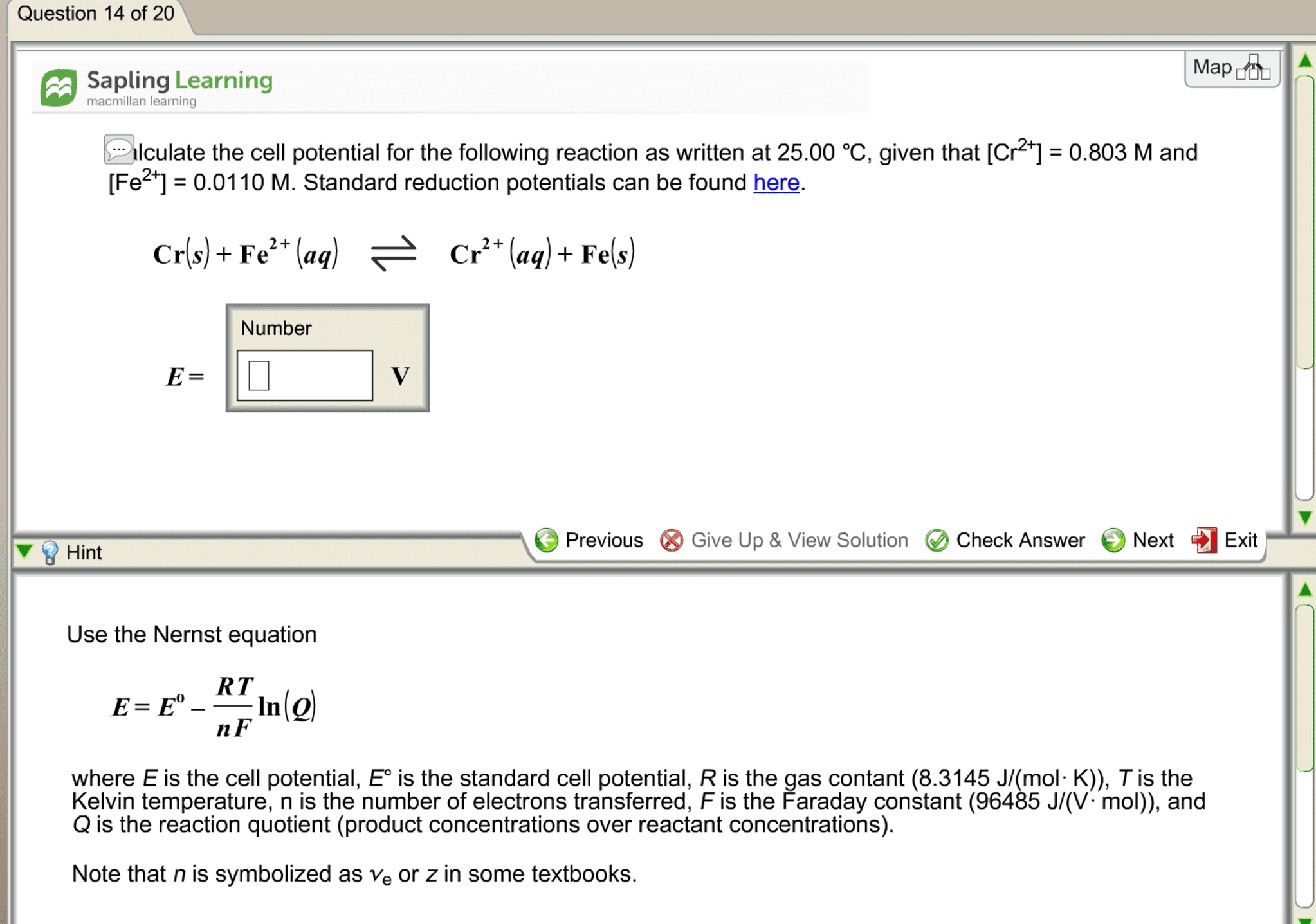
Lecture 9 notes: eo cell and thermodynamics, eo cell is directly related to go, the free energy change for the reaction. V = j/c: go = -nfeo cell. Where f = faraday constant: 9. 6485 104 j/v mol of e- (or 9. 6485 104 coulombs/mol of e-) And n is the number of moles of electrons transferred in the redox rxn. For a product-favored reaction: reactants products, go < 0 and so eo cell, eo cell is positive. When ecell = 0 (no net rxn), reactants and products are at equilibrium and q = k. 0 cell: nernst equation, electrolysis, using electrical energy to produce a nonspontaneous chemical change, electric energy chemical change (nonspontaneous, electrolysis of molten nacl, here a battery pumps electrons from cl- to na+. Note: polarity of electrodes is reversed from batteries (voltaic): electrolysis of h2o. Electrolyte added to pure h2o; e. g. na2so4 or dil acid.