CHM 2046C Lecture Notes - Lecture 4: Conjugate Acid, Acid Strength, Acid Dissociation Constant
Document Summary
Get access


Related Documents
Related Questions
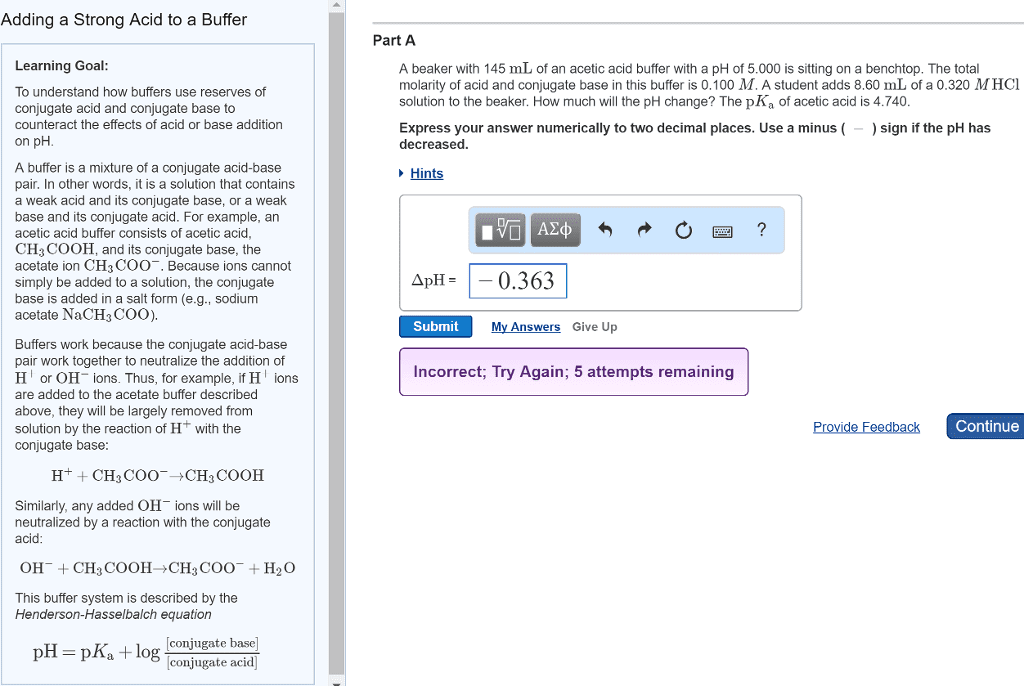
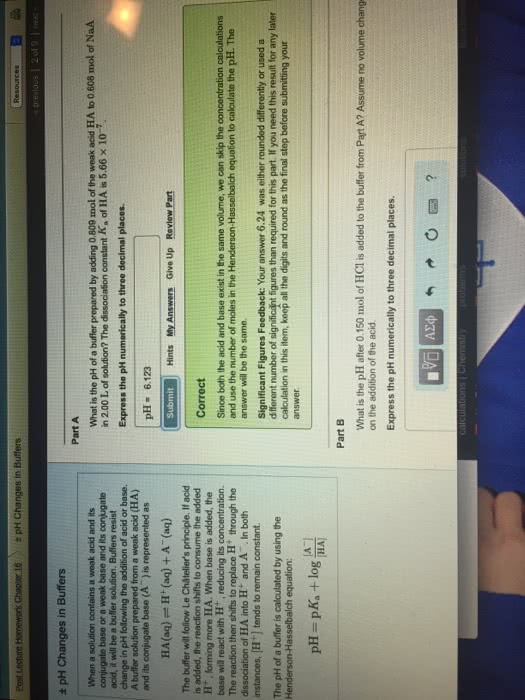
| ± pH Changes in Buffers When a solution contains a weak acid and its conjugate base or a weak base and its conjugate acid, it will be a buffer solution. Buffers resist change in pH following the addition of acid or base. A buffer solution prepared from a weak acid (HA) and its conjugate base (Aâ) is represented asHA(aq)âH+(aq)+Aâ(aq) The buffer will follow Le Châtelier's principle. If acid is added, the reaction shifts to consume the added H+, forming more HA. When base is added, the base will react with H+, reducing its concentration. The reaction then shifts to replace H+ through the dissociation of HA into H+ and Aâ. In both instances, [H+] tends to remain constant. The pH of a buffer is calculated by using the Henderson-Hasselbalch equation: pH=pKa+log[Aâ][HA] | Part A What is the pH of a buffer prepared by adding 0.405 mol of the weak acid HA to 0.609 mol of NaAin 2.00 L of solution? The dissociation constant Ka of HA is 5.66Ã10â7. Express the pH numerically to three decimal places. Hints
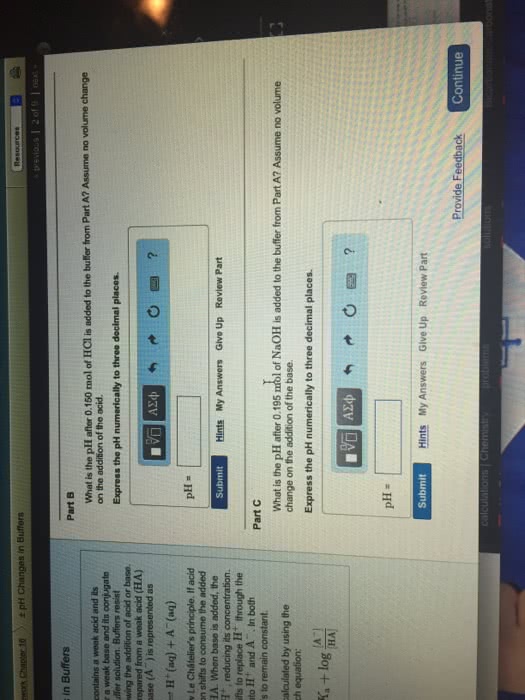
SubmitMy AnswersGive Up Correct Since both the acid and base exist in the same volume, we can skip the concentration calculations and use the number of moles in the Henderson-Hasselbalch equation to calculate the pH. The answer will be the same. Part B What is the pH after 0.150 mol of HCl is added to the buffer from Part A? Assume no volume change on the addition of the acid. Express the pH numerically to three decimal places. Hints
All attempts used; correct answer displayed Even though a significant amount of a strong acid was added, the pH of the buffer changed very little. If this amount of acid was added to 2.00 L of pure water, the pH would change from 7.000 to 1.125. Part C What is the pH after 0.195 mol of NaOH is added to the buffer from Part A? Assume no volume change on the addition of the base. Express the pH numerically to three decimal places.
Incorrect; Try Again; 4 attempts remaining |