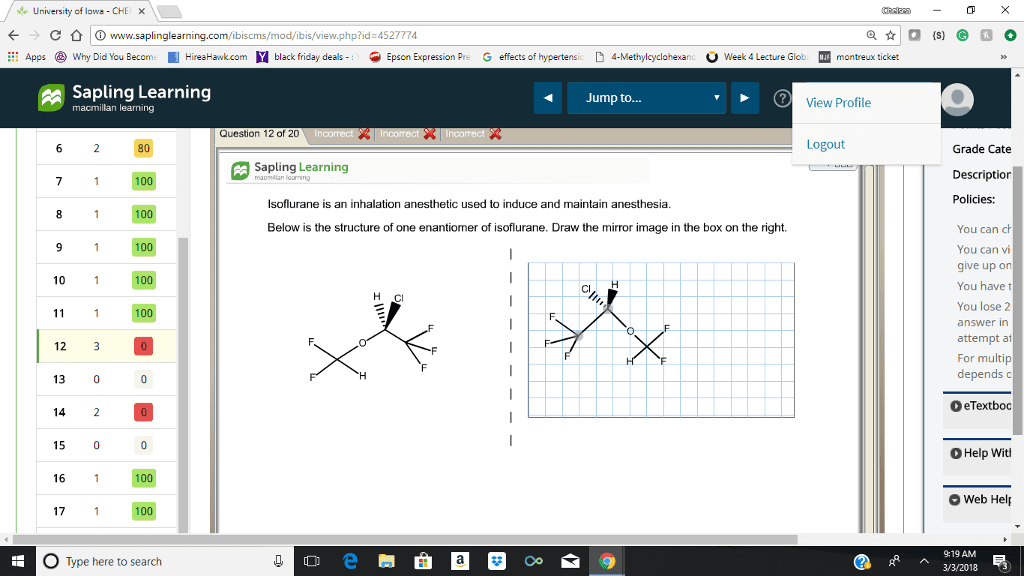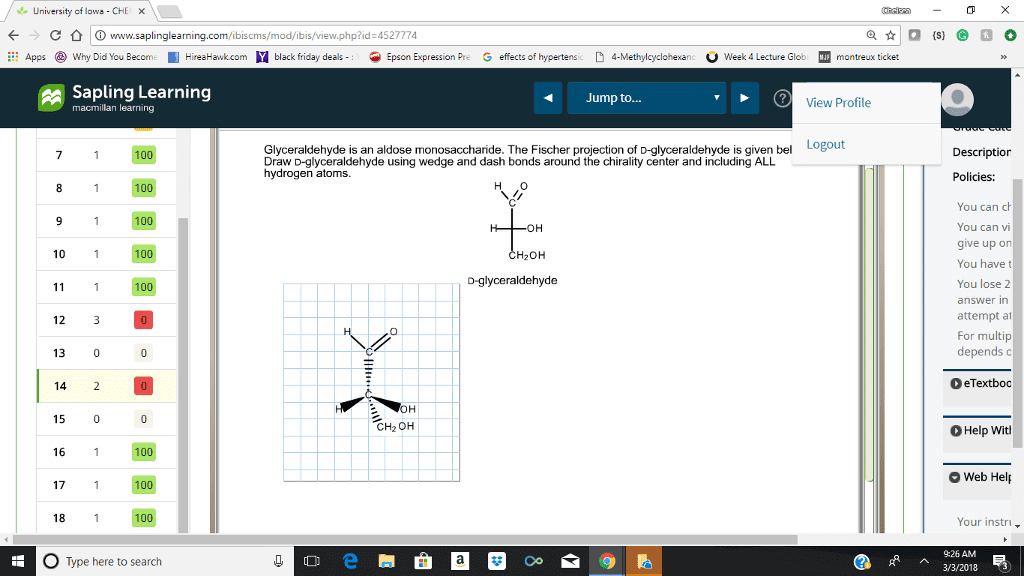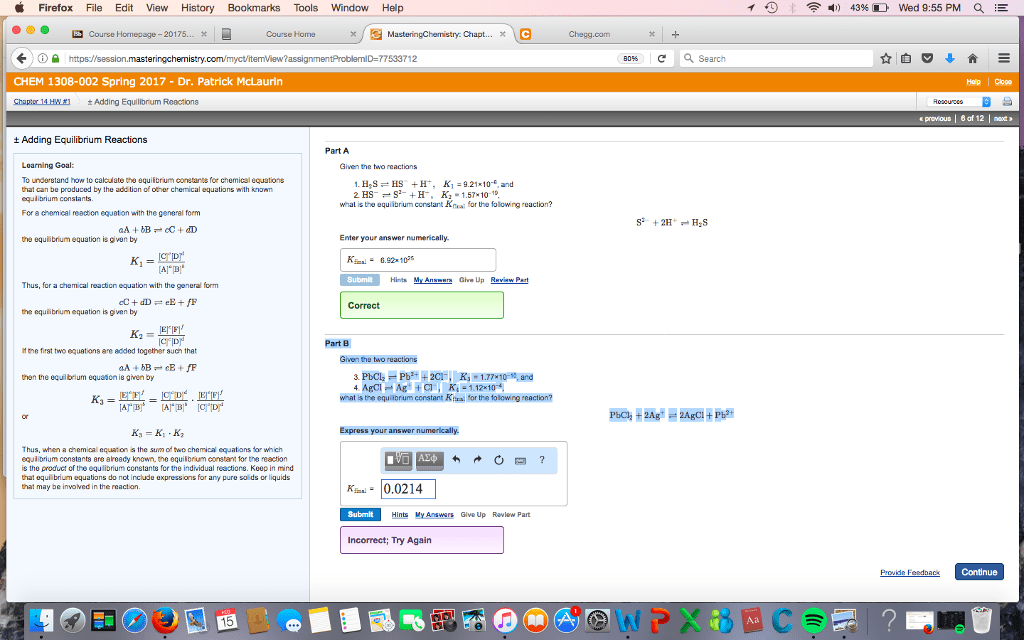
Step by step solution for part B would be great! Not sure what i am doing wrong
Firefox File Edit View History Bookmarks Tools Window Help Home x Masteringchemistry: Chapt... x VC Homepage-20175 https://session masteringc CHEM 1308-002 Spring 2017 Dr. Patrick McLaurin t Adding Equilibrum Reactions Adding Equilibrium Reactions Part A Loaming Goal Given the two To understand how to calculate the eq rum constants for chemical equations HSS S +H 9.2 E. and be produced by the addition of other chemical equatio with known 2 HS S H Ka 1.57 10-15 what is the equilibrium constant K for the folowing reaction? For a chemical reaction equaton with the general form Enter your answer numerically. the eq iibrium equation is given by chemical equation with the genera form cC+ dD Correct librium equation is gven by the eq CTID Part B Mthe first two equations are added together 3uch tha Given the two aA bB 201 then the equilibrium equation given by what s the equilibrium constant Keralfor the falowing reaction? press your answer numerically. of two chemical equatio for which equilibrium constants are aready known the eq rum constant for the reaction is the product of the equi m constante for the individual reactions. Keep mind that eq uilibri for any pure soid 0.0214 Submit Hints My Answers Give Up Review Part correct Try Again a search 2H Has 2AgC T 4) 43% Wed 9:55 PM E 6 of 12 Continue Provide Feed