Organic Chemistry lab QUESTIONS Extraction of Caffiene Lab
â¢Procedure
1) Use hot water to extract the caffeine from the tea leaves.
- tannins and chlorophyll will extract along with caffeine
2) Use dichloromethane to extract caffeine from water using extraction
techniques from last weeks lab.
- dichloromethane is more dense than water and will be the
bottom layer in the separatory funnel.
- perform 3 x 20 mL extractions with dichloromethane
3) Wash the 2 x 20 mL 6M NaOH solution with base to remove acidic
tannins. Tannins will deprotonate and preferentially partition into
the aqueous phase.
4) Dry organic phase with drying agent (CaCl2) to remove residual water.
5) Transfer dichloromethane to flask and evaporate in hood.
6) Recrystallize caffeine from 2mL hexanes and 0.5 mL acetone. Filter, mass product and determine % by weight of caffeine from tea.
7) Analyze for purity via TLC and compare to that that extracted from coke and pure caffeine..

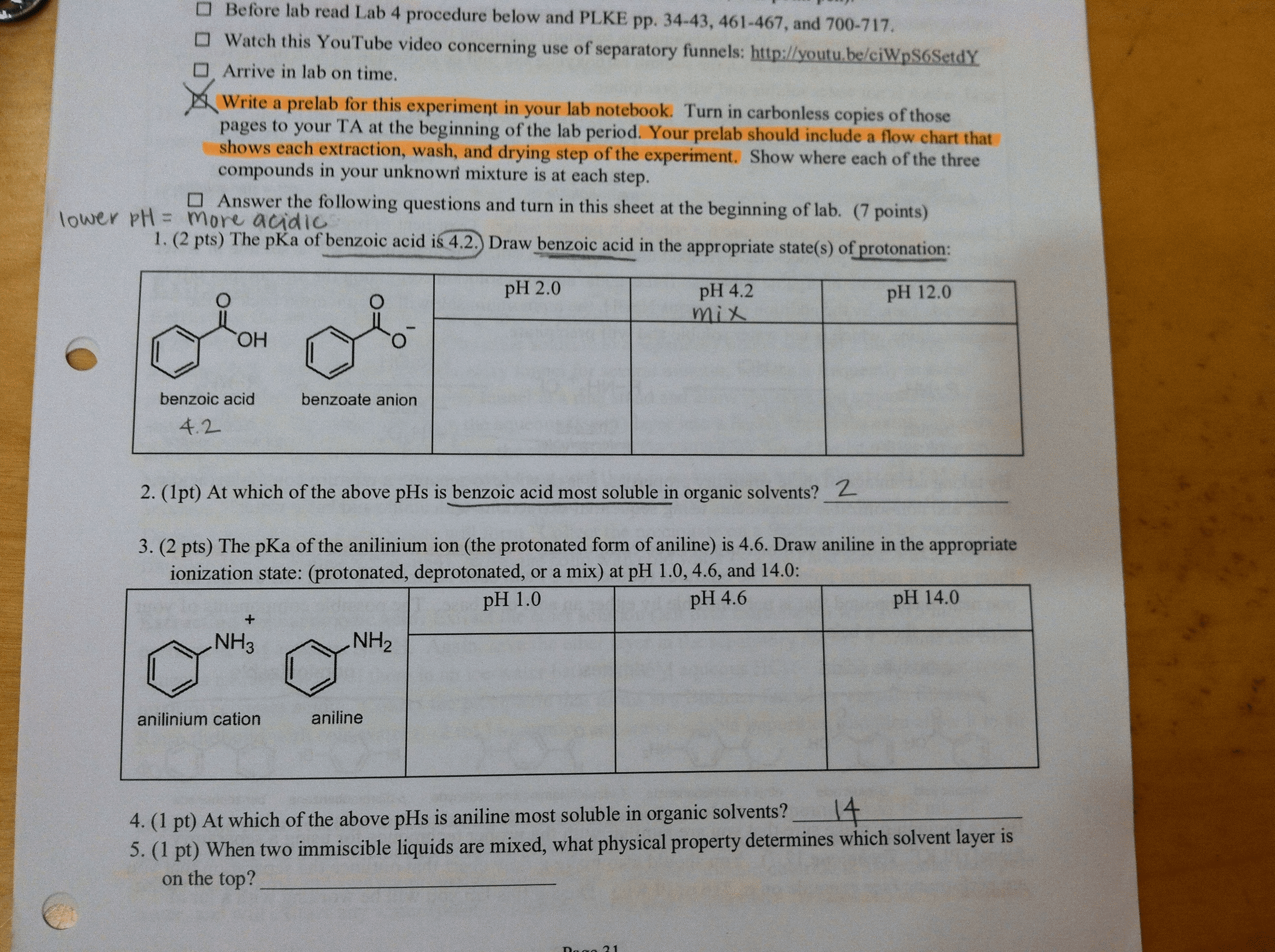
Post Lab Questions 1.(5pts) Prepare a flow chart for the experiment. Remember to include appropriate structures and explicitly state what compounds are present and in which layer/fraction for each step 2.(5pts)Other than relative solubilities, what are two additional factors that may be worth taking into consideration when choosing an organic solvent for use in a liquid-liquid extraction against water? Justify your choices. 5. (5pts) Percent recovery and percent yield are occasionally used as if they are interchangeable terms. They do, however, have different meanings. Why is it more correct to use percent recovery here? 6 (5pts) Would the apparent percent recovery be higher, lower, or unchanged ifa student did the following (answers must provide an explanation founded in the underlying chemistry) a. Used room temperature water to brew the tea? b. Did not add calcium chloride when directed?