CHEM 1124Q Lecture Notes - Lecture 5: Vsepr Theory, Sigma Bond, Lewis Acids And Bases
Document Summary
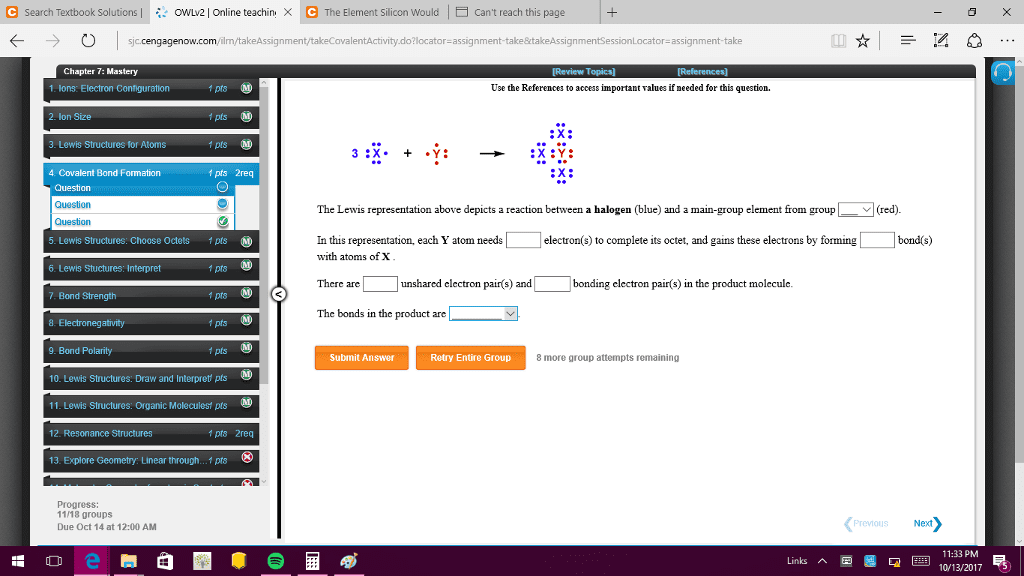
Bonds can be formed from one, two or three pairs of electrons. The order of a bond is the number of pairs shared: single bonds (1), double bonds (2), triple bonds (3) There are two extremes in the spectrum of bonding: covalent bonds occur between atoms that share electrons, ionic bonds occur between a metal and a nonmetal and involve ions. Bonds that fall between these extremes are polar. The delta, , is used to denote partial charges on the atoms. M:x, pure covalent bond, neutral atoms held together by equally shared electrons. M +x , polar covalent bond, partially charged atoms held together by unequally shared electrons. M+x , ionic bond, oppositely charged ions held together by electrostatic attraction. In polar covalent bonds, electrons are shared but not shared equally. Electron density maps show the distributions of charge. Electrons spend a lot of time in red and very little time in blue.