CHEM 6A Lecture Notes - Lecture 3: Molar Mass, Molecular Mass, Empirical Formula
36 views6 pages
30 Aug 2016
School
Department
Course
Professor


19
CHEM 6A Full Course Notes
Verified Note
19 documents
Document Summary
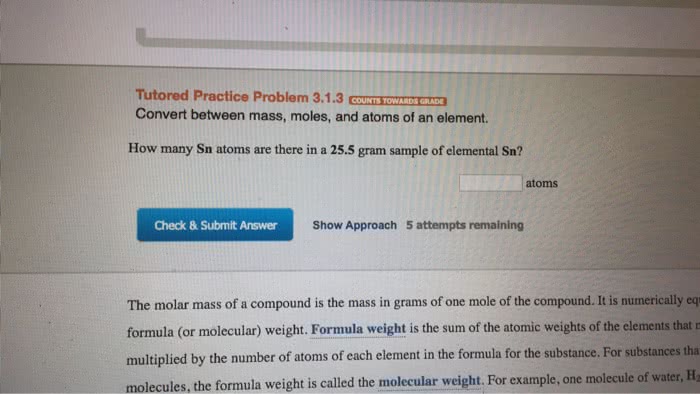
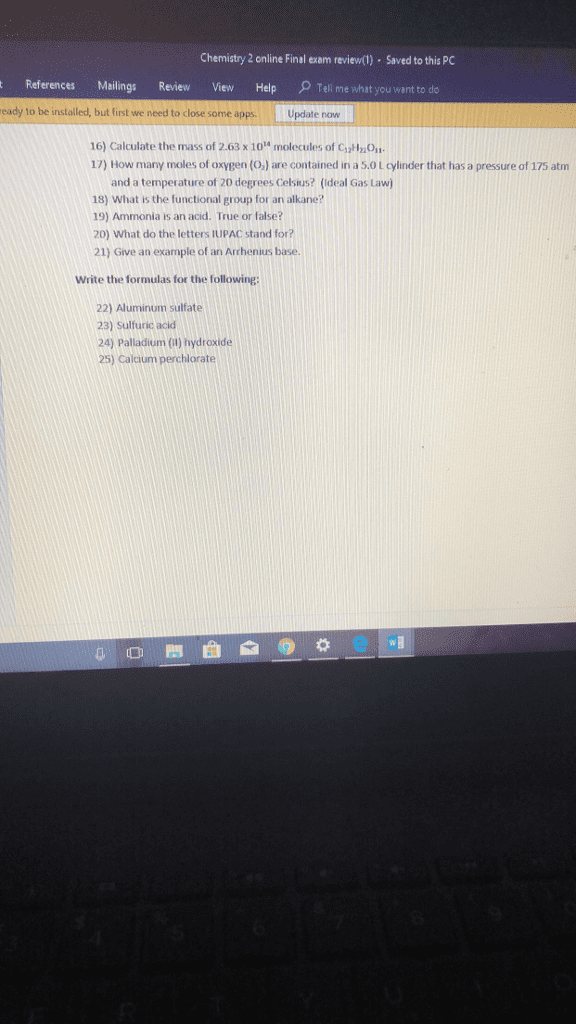
Molecular mass is the sum if all the masses of all atoms in a molecule: measured in amu. Formula mass is the sum of the masses of all atoms in a formula unit of an ionic compound: formula unit is the combination of numbers and chemical symbols used to define a molecule, measured in amu. Molar mass is the mass of one mole of atoms, ions, molecules, or formula units: measured in grams. Avogadro"s number: 6. 022 x 10^23, equals 1 mole, makes working with large numbers easier. Percent composition: the percent composition of a compound is a statement of the relative mass each element contributes to the mass of a compound as a whole, calculations: Convert each percent to grams by assuming 100 grams total. Divide each by individual molar mass to find number of moles of each element. Multiply by common multiplier until whole number.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232