CHEM103 Lecture Notes - Lecture 11: Flame Test
Document Summary
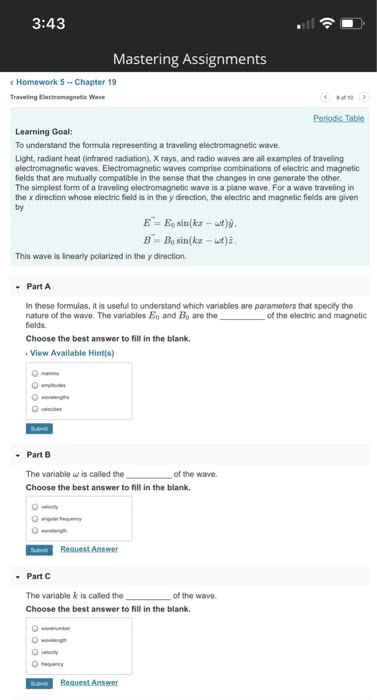
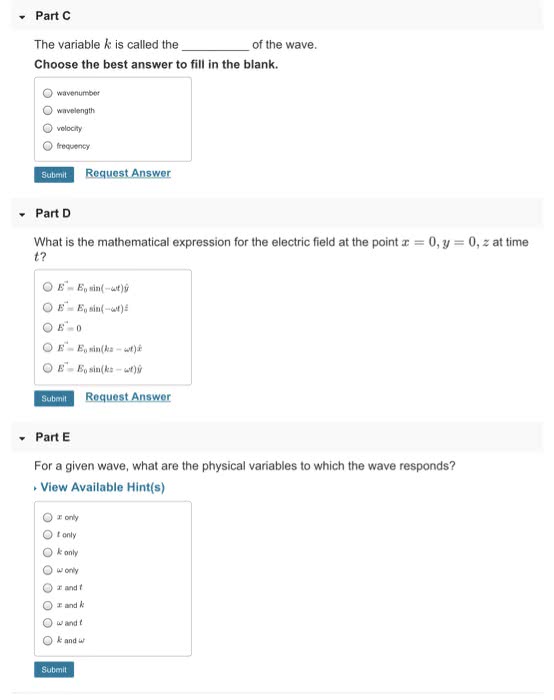
Chapter 7: light is a form of electromagnetic radiation, all electromagnetic waves move at speed of light. Frequency: number of waves that pass at point in each period, number of waves also called number of cycles, units are hertz (hz) or cycles/s = s-1. 1 hz = 1 s-1: how to calculate frequency. Spectra: some atoms and molecules absorb energy and then release it. Flame test where we put chemical in a flame and the flame changed colors. Photoelectric effect: many metals emit electrons when light shines on surface, to re(cid:373)ove a(cid:374) ele(cid:272)tro(cid:374), (cid:373)ust e(cid:454)(cid:272)eed ele(cid:272)tro(cid:374)"s (cid:271)i(cid:374)di(cid:374)g e(cid:374)erg(cid:455) Determine energy of a wave: higher amplitude. Waves with higher amplitudes should mean more electrons being emitted: brighter light should kick electrons out quickly. Dimmer light should take some time before enough energy is transferred. Magnetic quantum number: orientation of orbital. Values are integers from l to +l. Gives number of orbitals of a particular shape.