CHEM214 Lecture Notes - Lecture 1: Glutamic Acid, Aspartic Acid, Asparagine
Document Summary
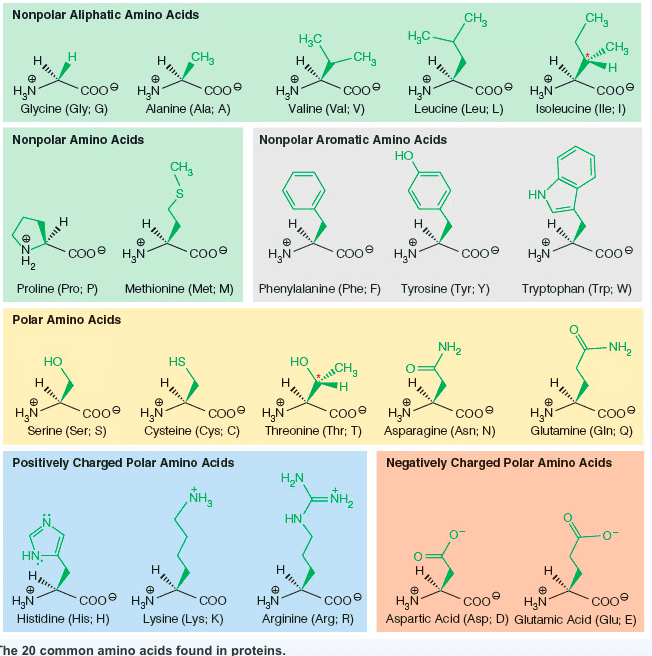
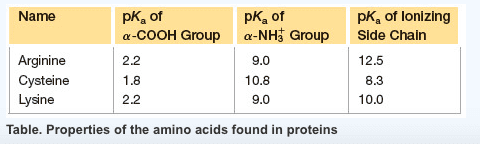
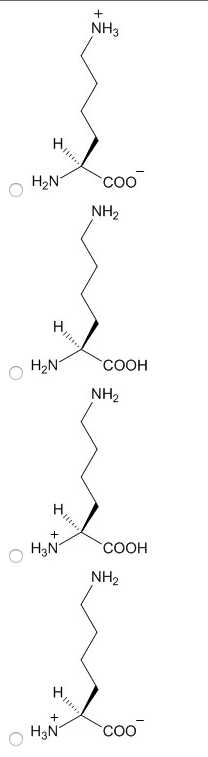
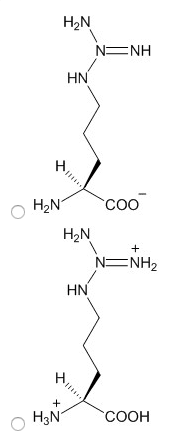
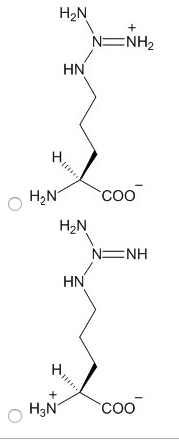
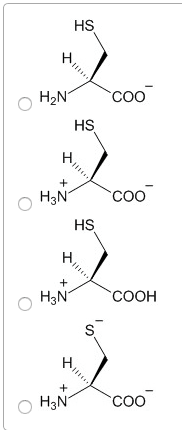
Characteristics: aspartic acid (proton on) and aspartate (proton off, glutamic acid (proton on) and aspartic acid (proton off, have measurable pka"s, only donate proton at high ph, negative at physiological ph, polar. Amino acids: aspartate (asp, polar, glutamate (glu, polar. Characteristics: have an amid group, are not ionizable, polar. Amino acids: asparagine (asn, polar, glutamine (gln, polar. Proteins: protein fold is based off of its hydrophobicity. Synthetic protein: the more carbons, the more hydrophob ic. We need to know: names of amino acids, amino acid abbreviations, what kind of r group each amino acid has, structures of the 6 amino acids, pka"s of n and c terminus, carboxylic acid, amine. Amino acids pt 2: ionization states of amino acids. Charge on a protein steps: draw all protons on, evaluate at ph given, add all charges. Peptides 1-degree structure determination: determining primary sequence because it dictates functionality, family of protein trees have similar sequences.