CHEM321 Lecture Notes - Lecture 1: Valence Electron, Bohr Model, Unified Atomic Mass Unit
Document Summary
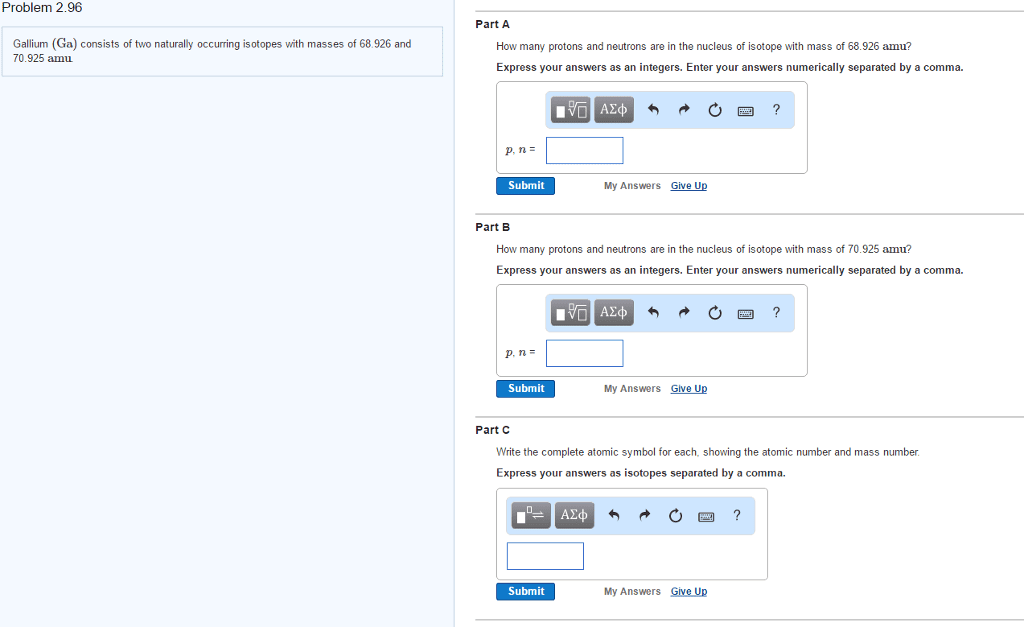
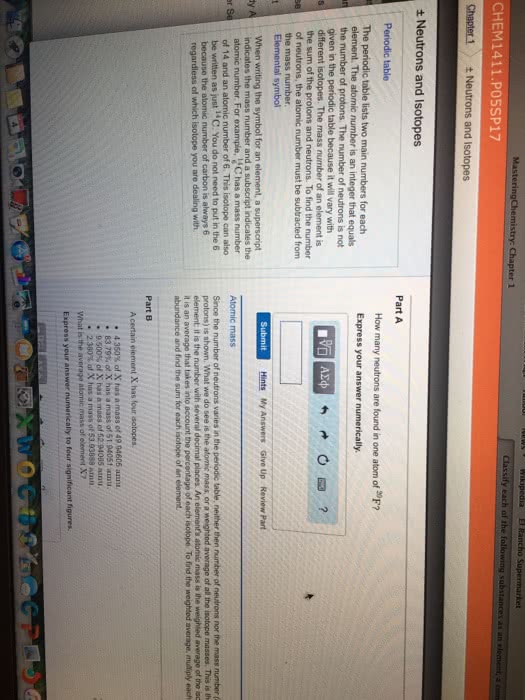
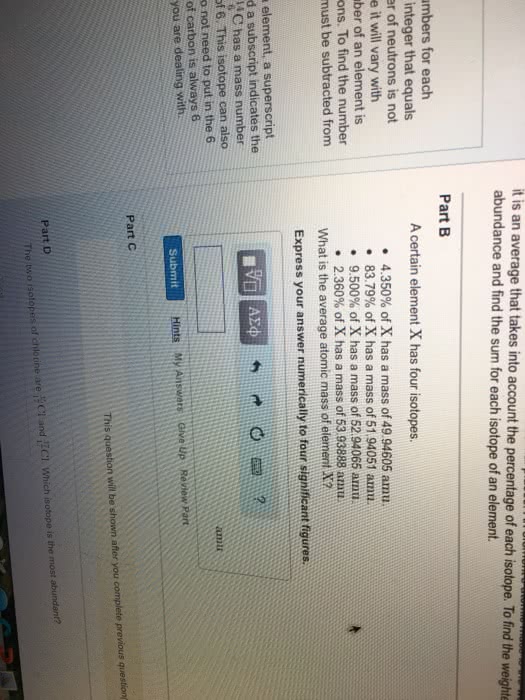
Is anything that takes up space and has mass. Problem of infinite divisibility is atoms are uncuttable. Cations are positively charged and anions are negatively charged. Ex: carbon has an atomic # of 6 meaning it has 6 protons, it has a mass # of 12, 12 6 = 6 n0. Another form of a certain element that has the same number of protons but a different mass due to an increase or decrease in neutrons. The mass number found on the periodic table for an element is an average of all of its naturally occurring isotopes. The bohr model was like a planetary model with the nucleus at the center and electrons in circular orbits around it. The energy an electron is storing is relative to its distance from the nucleus. The closer to the nucleus the more energy it takes to remove it. Valence electrons: are those found on the outermost orbital.