CHM 2045 Lecture Notes - Lecture 25: Haber Process, Transition Metal, Rate Equation
Document Summary
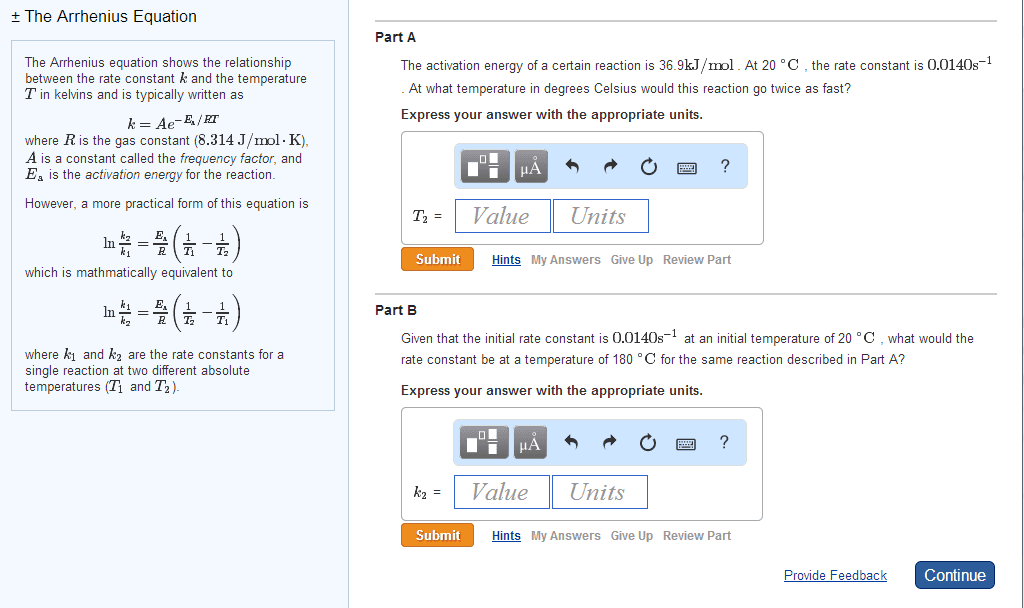
E(cid:894)ea) is amount of energy required to initiate a chemical reaction. K : temperature of reaction(t, activation energy(ea, frequency of effective collisions, orientation of colliding reactants ln(k) = (-ea/r) x (1/t) + ln[a] Overall progress of a chemical reaction can be represented at a molecular level by series of simple elementary reactions (steps) The sequence of elementary steps that leads to reaction formation is reaction mechanism. Intermediates species that appear in a reaction mechanism but not the final equation. Intermediates are always formed early and consumed in the later step. The molarity of a reaction depends the number of molecules reacting in an elementary step. Termolecular reaction elementary step with three molecules the slow step determines the rate law. The experimental rate law for the reaction between no2 and co to produce no and co2 is rate = k[no2]2. The reaction is relieved to occur via two steps: Catalyst substance that increases the rate of reaction without being consumed.