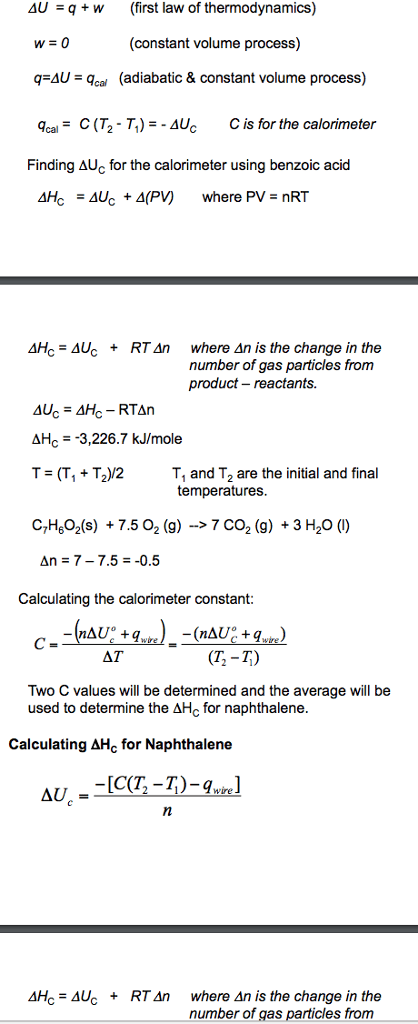Calorimerty and enthaply oof combution lab.
Attention: I need help with math cad. Please dont answer it anyother form. If you need the mathcad software email me. khan 11 27 94 @ g m a i l dot com. i will compansate you.
LAB DATA:
Benzoic acid #1
Temp: Temp inital =21.2 deg temp final 23.6
Mass: .9961 Grams.
Wire: 9.3 cm..... all wire were used.
Benzoic acid #2
Temp: Temp initial=22.1 Temp final 24.7
mass: 1.0137grams
wire: 9.3 cm....... .8cm left
Napthalene #1
Temp: Temp initial=21.8c...Temp final=25.6c
Mass: .98grams
Wire: 9.3cm inital 1.0cm left
Napthalene#2
T1= 21.9 T2=28.8
Mass=1.0777g
wire:9.3 to .5cm
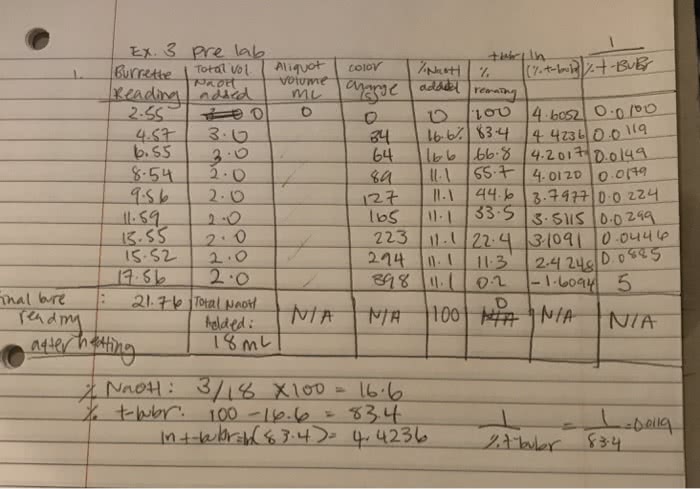
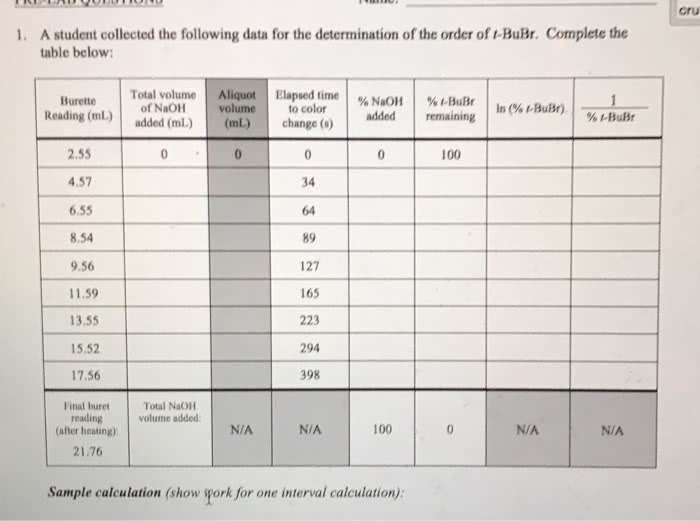
1.. Determine the calorimeter heat capacity using the known enthalpy of combustion of benzoic acid and from the equation below . 2. Calculate ÎHc for naphthalene using equations
. Again, be sure to correct for the heat of combustion of the ignition wire.
4. Use MathCad to determine the propagated errors in the calorimeter heat capacity, C, due to possible errors in the measurement of the temperature.
5. Calculate and compare the % error between your experimental heat of combustion of naphthalene and the literature value.
4U q W (first law of thermodynamics) (constant volume process) q AU cal (adiabatic & constant volume process) qcal E C (T TT) AU C is for the calorimeter Finding AUC for the calorimeter using benzoic acid 4HC -4Uc 4 (PV) where PV nRT 4HC 4Uc RT An where An is the change in the number of gas particles from product reactants. AU RTAn AH 3,226.7 kJ/mole T (T, T /2 T, and To are the initial and final temperatures. C7H6o2 (s) 7.5 O2 (g) 7 CO2 (g) 3 H2O (l) 0.5 Calculating the calorimeter constant wire AT (12-T) Two C values will be determined and the average will be used to determine the AHC for naphthalene Calculating AHC for Naphthalene AUC 4HC 4Uc RTAn where An is the change in the number of gas particles from