CHEM 1331 Lecture Notes - Lecture 1: Pipette, Photon
40 views2 pages
6 May 2016
School
Department
Course
Professor
Document Summary
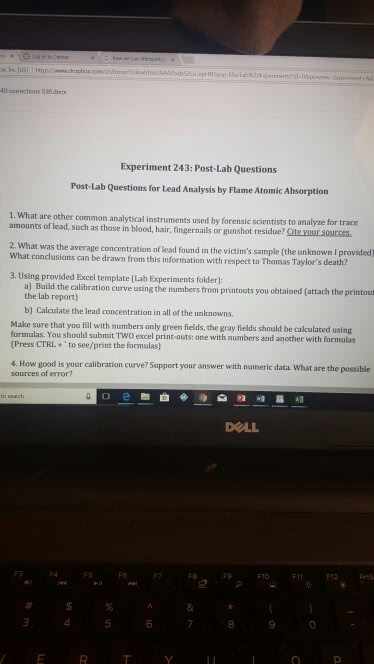
Include your name, date of the experiment and your drawer # in every lab report: write down the objectives for every experiment that you do, fill out all of the tables with your data and results. Always include units with your numbers: unless you told otherwise, include the analysis of your experiment (conclusions) in every lab report. Do not try to use your observations and/or calculation methods in the place of conclusions. The questions provided below should help you with it. Lab exercise # 1 techniques and measurements: compare the precision of the three balances used in mass measurements. Which one is the least precise: compare the precision of all volumetric glassware used in volume measurements (beaker, pipette, graduated cylinders, buret). Lab exercise # 3 density: parts 1-3 of your lab experiment involved determining the densities of various solids, liquids and gas.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232