CHEM 102 Lecture Notes - Lecture 21: Ionic Compound, Covalent Bond, Bond Length
Document Summary
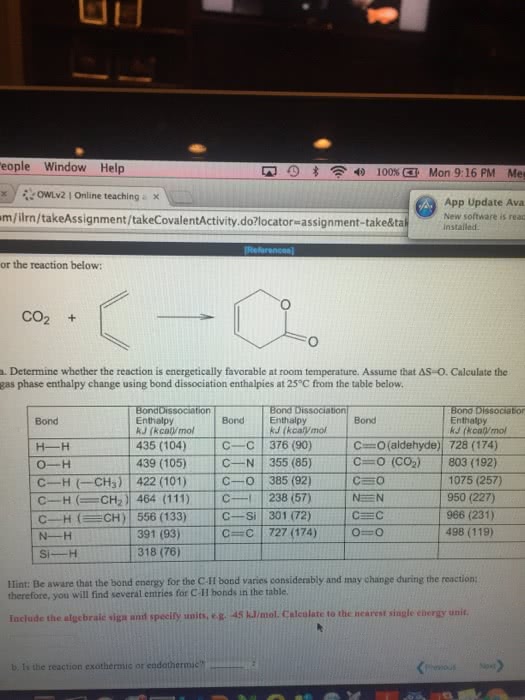
Energy required to break 1 mol of a covalent bond between atoms in a molecule in the gas phase. H = [2(436) + 498] [4(464)] = 486. Does not include imf"s (important for phsycial reactions) Energy required to melt 1 mol of a substance. H2o (s) --> h2o (l) hfus= 6. 02 kj/mol. Energy required to boil 1 mol of a substance. H2o (l) --> h2o (g) hvap = 40. 7 kj/mol. Energy required to sublimate 1 mol of substance. In order to freeze, just reverse the reaction and switch the signs of h. An ice cube tray is filled with 10. 0 g (0. 555 mol) of water at 27 c and placed in the freezer where the water freees and has a final temp. of -10 c. How much heat was freezer where the water freees and has a final temp. of -10 c. H2o (s) @ -10 c (10g)(4. 18)(-27 c) = -1128. 6 j.