CHEM 130 Lecture Notes - Lecture 8: Azimuthal Quantum Number, Pauli Exclusion Principle, Electron Configuration
Document Summary
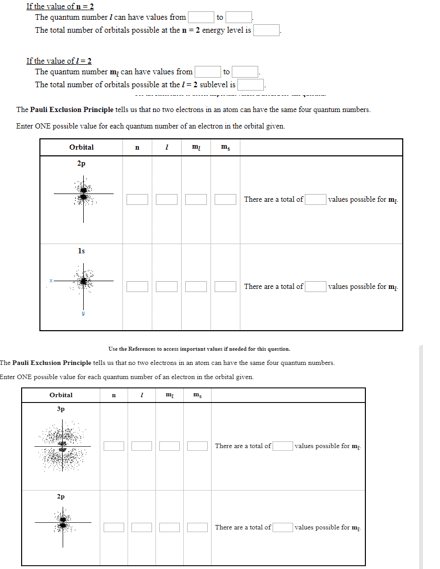
Note: we will only cover sections 8. 1 8. 6. 8. 1 electron spin and the pauli exclusion principle. As we learned in chapter 7, an electron can have a spin quantum number of + or - - this number represents the orientation of the magnetic spin axis. Ex: lithium has a configuration of 1s22s1, indicating that there are 2 electrons in the 1s orbital and 1 electron in the 2s orbital. The pauli exclusion principle states that no two electrons can have the same four quantum numbers. This means that two electrons in the same orbital (remember: an orbital is described by n, l, and ml) must have different spin quantum numbers. This also tells us that each orbital can only hold two electrons. The lowest energy state of an atom is called the ground state and corresponds to a particular electron configuration.