CHEM 111 Lecture Notes - Lecture 7: Sulfite, Oxalate, Thiocyanate
Document Summary
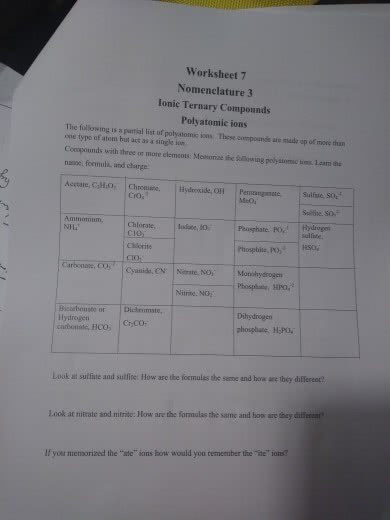
The naming of polyatomic ions is based on how many oxygen is in the formula: suffix: For main group elements, their opportunity to become a cation or an anion depends on the atoms electronegativity. Outer most shell is known as the valence shell. The first sphere will always have two electron. Atoms has the tendency to lose or gain electrons in order to achieve a full shell. Any group that is not a noble gas will lose or gain electron to achieve a full shell. Group 1,2, & 13 forms cations; charge = # of valence electrons or group # *if a group has fewer than 4 valence electrons: it is easier to lose electrons than to gain electrons. Could lose four electrons or gain four electrons. +4 is more common because less electronegativity allows it to lose their electron being easier than gaining electrons. No characteristic group charges many charges, many oxidation state.