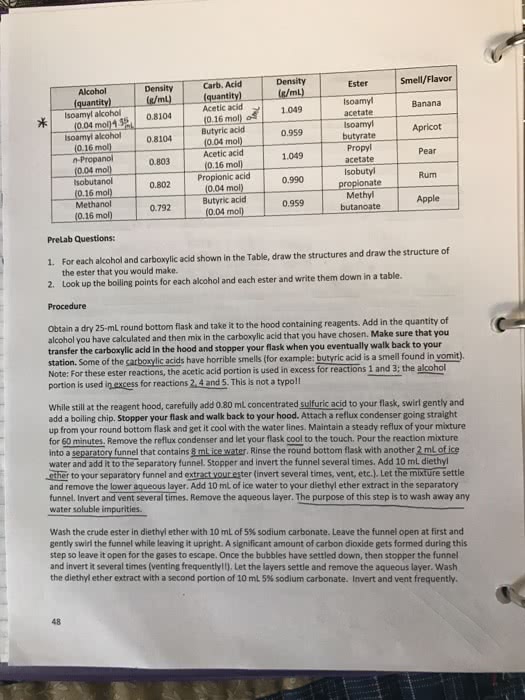
CHEM 250 Lecture Notes - Lecture 20: Hexanoic Acid, Carboxylic Acid, Butanone
15 views6 pages
Document Summary
The functional group of a carboxylic acid is a carboxyl group , which can be represented in any one of three ways. For an acyclic carboxylic acid take longest carbon chain that contains the carboxyl group as the parent alkane. Drop the final - e from the name of the parent alkane and replace it by -01c acid. Number the chain beginning with the carbon of the carboxyl group. Because the carboxyl carbon is understood to be carbon. 1 there is no need to give it a number. # oh noh hexanoic acid ( caproic acid) 3 - methyl butanone acid ( t sol valeric acid ) An - oh substituent is indicated by the prefix hydroxy. An - n hz substituent is indicated by the prefix amino - 4 - amino benzoic acid add the suffix - dioic acid to name of the parent alkane that contains both carboxyl groups ; thus ,
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232