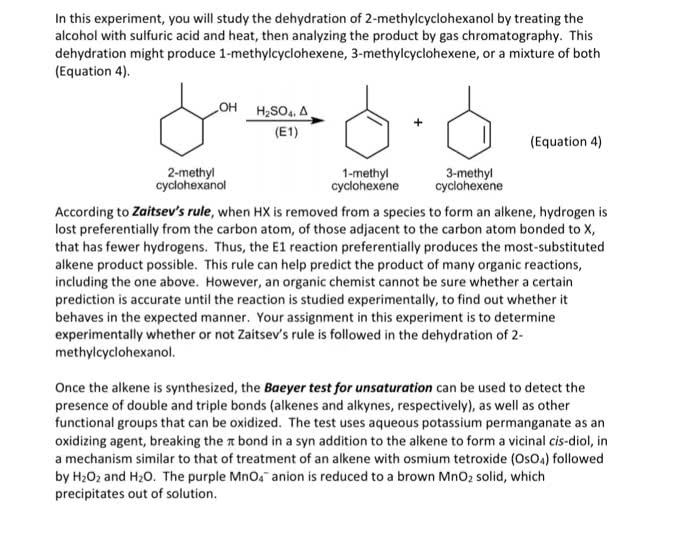
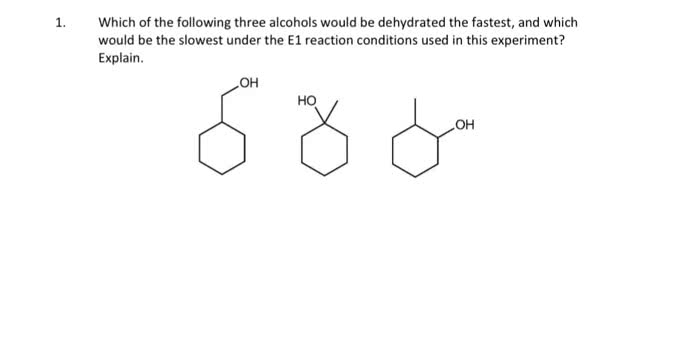
CHEM 261 Lecture Notes - Lecture 28: Lindlar Catalyst, Alkyne, Alkene
Document Summary
Chem261 lecture 28 alkyne reactions ii. Oxidative cleavage of alkynes: an internal alkyne treated with potassium permanganate or ozone will form two carboxyl groups, a terminal alkyne treated with potassium permanganate or ozone will form one carboxyl groups and carbon dioxide. Reducing alkynes: treating an alkyne with hydrogen and a catalyst will give an alkene and then an alkane, please note that the reaction does not stop at the alkene: it goes all the way to an alkane. Alkyne acidity: acetylide anions: a terminal alkyne treated with a cation and an amine group ionizes the alkyne. Acetylides as nucleophiles: now that you have an ionized alkyne, or an acetylide anion, you can treat it with. R-x (x=halogen, r=rest of the molecule) to attach the r group to the acetylide anion: the cation and halogen will form a salt, here is the breakdown of this mechanism, now let"s put it all together.