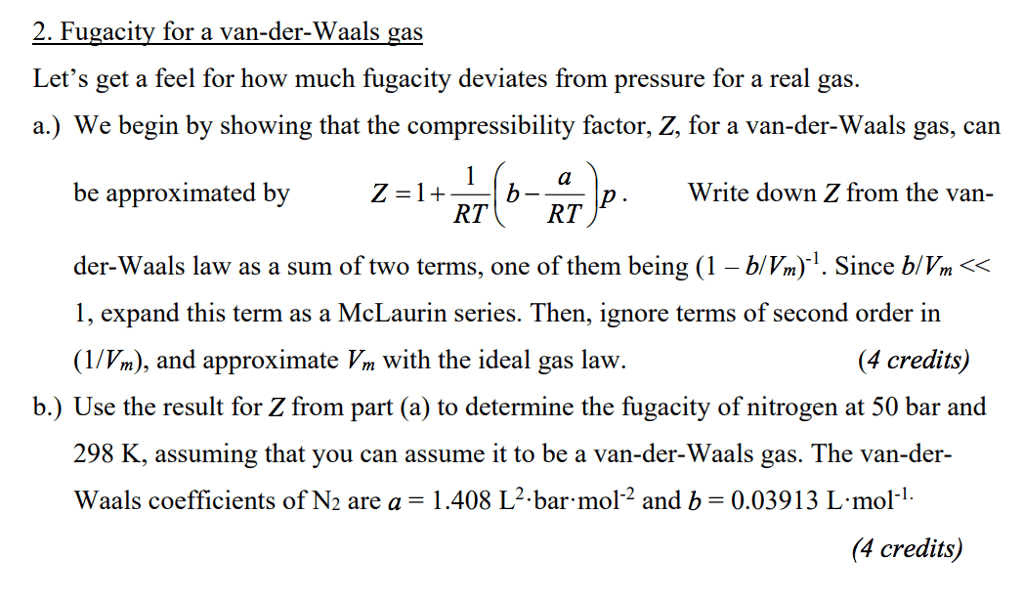CHEN 3101 Lecture Notes - Lecture 16: Dimensional Analysis, Ztt Records, Egg Cell
Document Summary
Recap: started w/ vander waals eqn of state. P [=] energy/volume thinking of rt as a variable rather than t. [=] volume/mol (b/c subtracting it from molar volume) a [=] (energy/mol) * (volume/mol) Van der waals is a generalized 2 parameter eos. Reltationship (to w/in prefactors) is given by dimensional analysis. Let z --> 1 for ideal gas (z has no dimension) can nondimensionalize t and p. Z3 + (-1-b)z2 + az - ab= 0. Chen thermo page 2 if you have a problem and you non-dimensionalize the solution, then has to depend on nondimensional. #s nondimensional v depends on the nondimensionalized p and t. If van der waals were exactly correct: (1) it would imply that z is a universal function of tr and pr (2) vdw eos would accurately describe this universal function z(tr,pr) (1) is much better than (2) Figure 6. 6-2 from textbook each line = isotherm.