CHEM 101 Lecture Notes - Lecture 8: Electron Configuration, Atomic Number, Alkaline Earth Metal
Document Summary
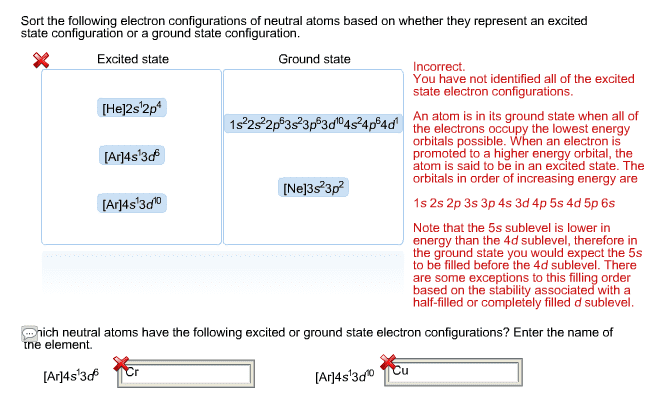
To deter(cid:373)i(cid:374)e the ele(cid:272)tro(cid:374) (cid:272)o(cid:374)figuratio(cid:374) for a(cid:374)y parti(cid:272)ular ato(cid:373), (cid:449)e (cid:272)a(cid:374) (cid:862)(cid:271)uild(cid:863) the stru(cid:272)tures i(cid:374) the order of atomic numbers. Each added electron occupies the subshell of lowest energy available. Electrons enter higher-energy subshells only after lower-energy subshells have been filled to capacity. For example, after filling the 3p block up to ar, we see the orbital will be 4s (k, The arrow leads through each subshell in the appropriate filling order for electron configurations. Simply make a column for all the s orbitals with each n shell on a separate row. Repeat for p, d, and f, like the illustration on the left. Finally, draw diagonal lines from top to bottom as shown. An alternate way to find write the electron configuration is by reading the periodic table. Ea(cid:272)h le(cid:448)el (cid:272)orrespo(cid:374)d to a period of the periodi(cid:272) ta(cid:271)le. Let"s take the groups a(cid:374)d di(cid:448)ide i(cid:374) (cid:271)lo(cid:272)ks as shown in the illustration below: