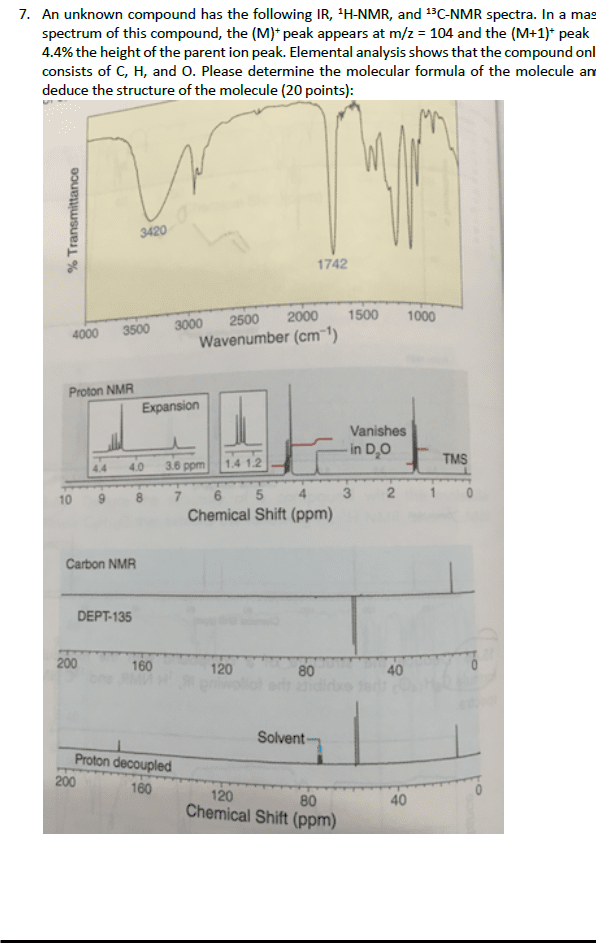
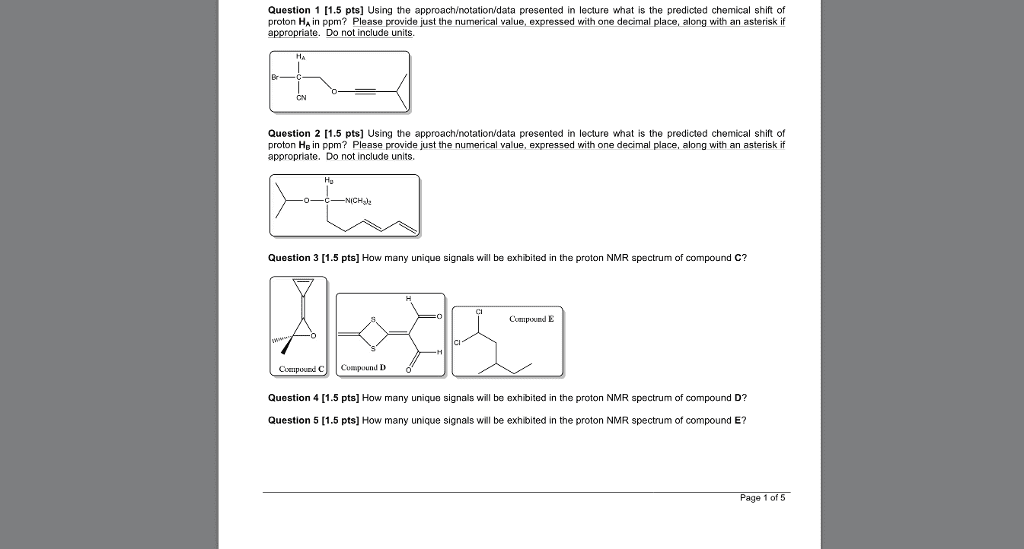
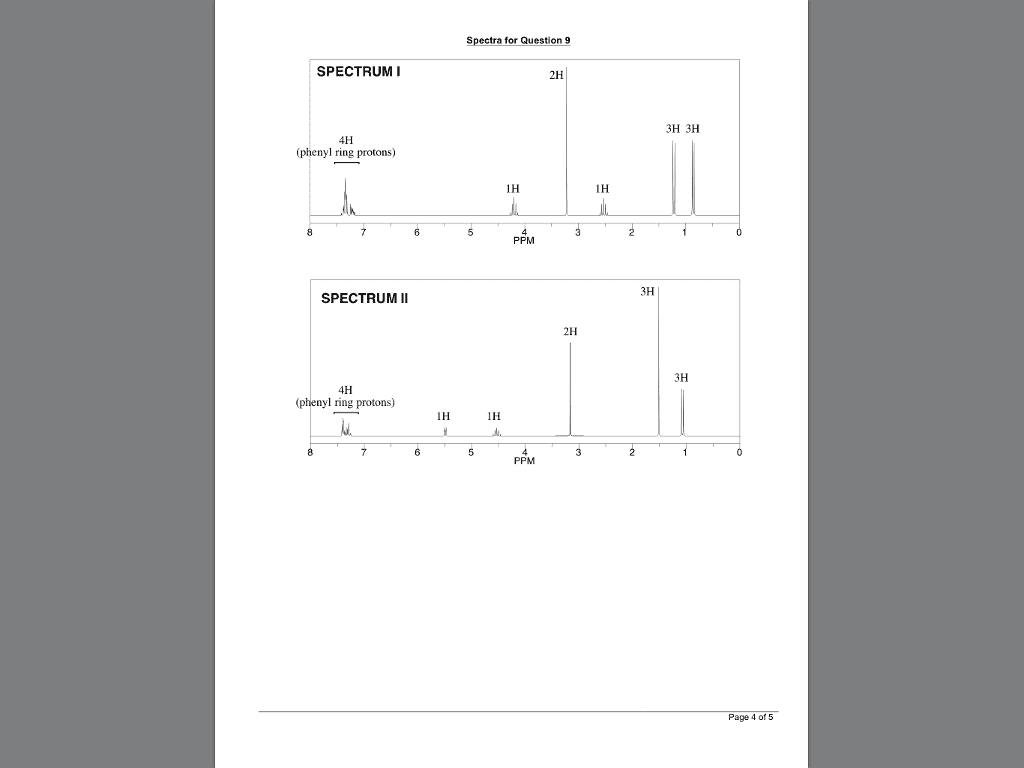
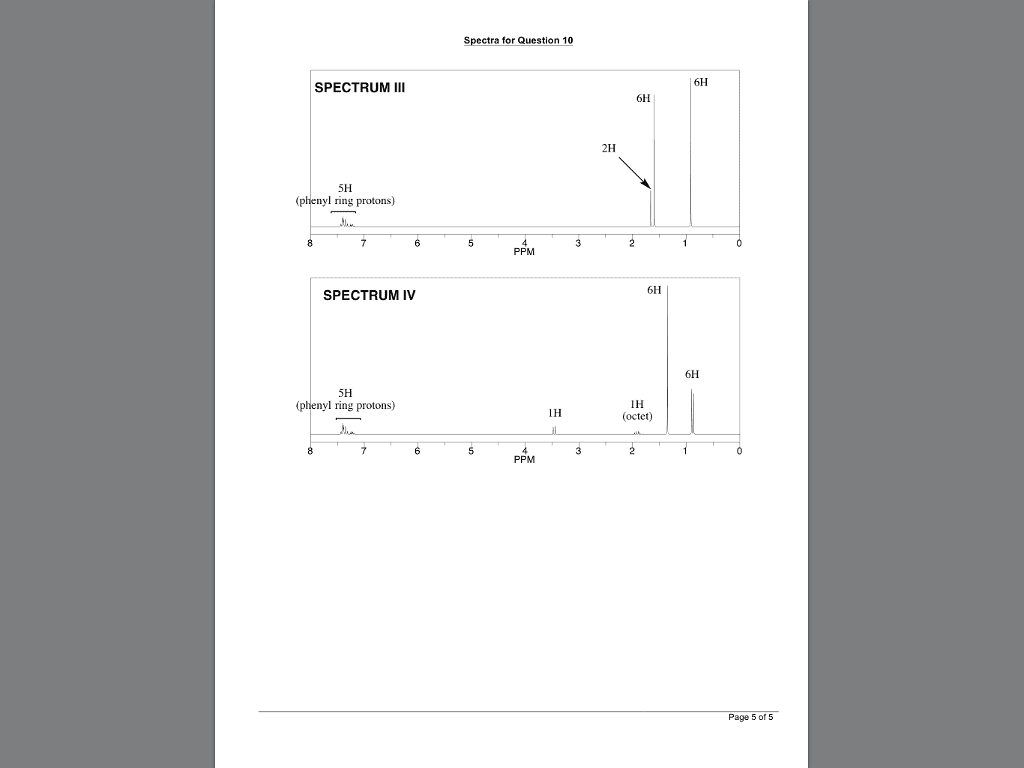
CHEM 2320 Lecture Notes - Lecture 12: Fiji, Chemical Shift, Diamagnetism
Document Summary
The electrons donated by oxygen can be found anywhere s. Se q a a causes shielding , up field goes along the ring . When you have a lot of branches in the carbon it shifts it downfield. The carbonyl carbon would be moved downfield partly because of the diamagnetic. Also more downfield due to e- withdrawing group . It add groups (4) that can donate electron density range . moving around fij. Within the carbonyl a can split the carbonyl groups into. ?dfj= to them like a double bond or a benzene ring . 8 baseline due to insensitive method . noisy b t to f. a t. ms. However there are three methyl groups that are. Three similar methyl groups so their peak must be taller. Can also identity them based on the number of peaks that arise due to symmetry. 11 so there must be some kind of symmetry.